We have treated over 3,500 Long COVID and Post-Vaccine Syndrome cases since 2022. However, still, the treatments showing the most promise in clinical practice aren’t yet in the official guidelines.
This isn’t surprising. The gap between clinical innovation and institutional approval often takes years, sometimes decades, to close. That is, if they ever close at all. There is, of course, story after story of promising curative treatments purchased by pharmaceutical companies and immediately thrown in the filing cabinet of oblivion – innovation killed and stifled to maintain cash cow products that produce no meaningful results.
Now infamous, the time a Goldman Sachs analyst asked the quiet part out loud: “Is curing patients a sustainable business model?” The analyst when on to write in her report, “…such (curative) treatments offer a very different outlook with regard to recurring revenue versus chronic therapies. While this proposition carries tremendous value for patients and society, it could represent a challenge for genome medicine developers looking for sustained cash flow.”
Long COVID and Post-Vaccine Syndrome patients can’t wait to see if someone looks favorably at the economic viability of helping them with their severe and debilitating conditions. And if that endeavor is decidedly profitable (which it most certainly will be based on the current trajectory of severely ill spike protein patients), they can ill afford to wait the additional 5+ years for clinical research and testing for expensive, patented drugs that treat symptoms.
What follows is an evidence-based discussion of a small handful of treatments we’ve used successfully in clinical practice, why they work mechanistically, what the research shows, and why most physicians haven’t adopted them yet. This is by no means an exhaustive list. But, some treatments we thought were worth highlighting for one reason or another.
This represents real-world clinical experience combined with emerging research. The kind of information you need to have informed conversations with your healthcare provider.
Understanding the Treatment Gap
Before discussing specific treatments, it’s important to understand why there’s often a delay between what helps patients and what becomes “standard of care.”
Three Primary Barriers:
1. Regulatory Framework Most of these treatments are FDA-approved for other conditions but used “off-label” for Long COVID. While legal and common in medicine, off-label use requires physician comfort with clinical decision-making beyond established protocols. We saw during the pandemic that “off-label” suddenly became a dirty word, weaponized against certain drugs demonstrating promise in treatment. More on that later.
2. Evidence Timeline Rigorous clinical trials take 3-5 years from design to publication. Long COVID emerged in 2020. We’re only now seeing results from the first wave of controlled trials. Furthermore, these trials are often on patented therapies with high side effect profiles and little clinical efficacy.
3. Risk-Benefit Calculation Physicians must weigh potential benefit against professional liability, especially for treatments lacking specific FDA approval for Long COVID. Even when clinical rationale is strong. To us, the decision is easy. Integrity trumps all risk.
The result: Patients often wait years for treatments that clinical experience suggests could help now.
Treatment #1: Low-Dose Naltrexone (LDN)
What it is: Naltrexone at standard doses (50-100mg/day) treats opioid addiction. At lower doses (1-4.5mg/day), it functions as an immunomodulator with anti-inflammatory properties.
Clinical rationale for Long COVID:
Low-dose naltrexone works through multiple mechanisms relevant to Long COVID pathophysiology:
- Immune modulation: Inhibits pro-inflammatory toll-like receptor 4 (TLR4), which drives cytokine production
- Cytokine reduction: Decreases inflammatory markers including IL-6, TNF-alpha, and IL-1β
- Ion channel restoration: A 2025 study demonstrated LDN restored TRPM3 ion channel function in Long COVID patients’ immune cells—significant because TRPM3 dysfunction impairs cellular energy production and immune response
- Mast Cell Stabilization: By reducing immune reactivity and cytokine production, it also creates a more stabile environment, preventing histamine release from mast cells.
- Neuroinflammatory Reduction: Reduces microglial activation in the brain, with 60% of users reporting reduction in anxiety, fatigue, and brain fog
Current evidence:
The NIH RECOVER program selected LDN for clinical trials, with enrollment beginning summer 2026. Existing data includes:
- Meta-analysis of observational studies (n=155 across 4 studies): Moderate effect size for fatigue reduction (Hedges’ g = -0.74, p<0.001)
- Quality of life improvements: SF-36 scores increased from 36.5 to 52.1 (p<0.0001) over 12 weeks
- TRPM3 restoration study (Frontiers in Molecular Biosciences, 2025): Demonstrated objective improvement in immune cell calcium signaling
- Safety profile: Excellent across multiple studies. Most common side effects are mild and transient (vivid dreams, slight headache in first 1-2 weeks)
Contraindications: Active opioid use, liver dysfunction
Clinical experience: In our practice, approximately 50-60% of Long COVID and Post-Vaccine Syndrome patients experience significant benefit from LDN. Response is most notable for fatigue, post-exertional malaise, and brain fog.
Typical response timeline: 8-12 weeks at therapeutic dose.
Prescribing approach:
- Start low (0.5-1mg) to assess tolerance
- Titrate slowly: increase by 0.5mg every 3-5 days
- Target dose: 3-4.5mg daily
- Taken at bedtime (may initially cause vivid dreams)
- Requires compounding pharmacy
Why adoption is limited: Requires off-label prescribing, compounding pharmacy access, and physician comfort with immunomodulation. Many physicians prefer to wait for RECOVER trial results before prescribing. To us, it is shocking that it has taken this long to even get studies going on this low risk, high benefit option.
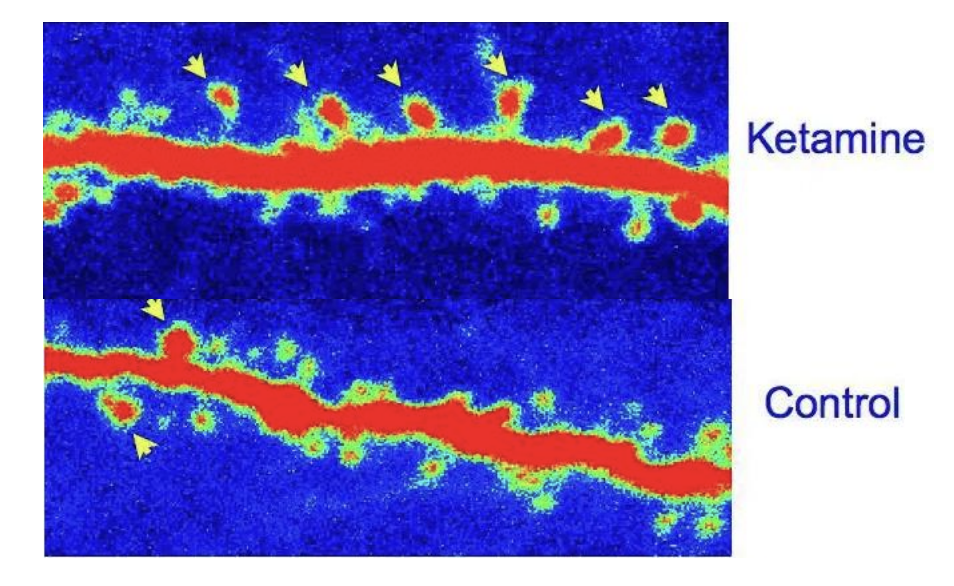
Treatment #2: Low-Dose Ketamine
What it is: Ketamine is an NMDA receptor antagonist originally used for anesthesia, now FDA-approved for treatment-resistant depression via the esketamine nasal spray (Spravato). We have written more extensively about its history, its mechanisms of action, and what we’ve seen in our Long Covid and Post-Vaccine Syndrome patients here
Clinical rationale for Long COVID and Post-Vaccine Syndrome:
Ketamine addresses multiple Long COVID and PACVS mechanisms:
- Neuroinflammation reduction: Downregulates inflammatory cytokines (IL-6, IL-17A, TNF-alpha) that contribute to brain fog and cognitive dysfunction
- Neuroplasticity enhancement: Increases BDNF (brain-derived neurotrophic factor), promoting neuronal recovery from COVID-related damage
- Rapid-acting antidepressant: Unlike traditional SSRIs requiring 4-6 weeks, ketamine can show effects within hours to days, and takes a fundamentally different approach from serotonin based treatments
- Chronic pain modulation: Effective for neuropathic pain through NMDA receptor antagonism
Current evidence:
- Active clinical trials: University of British Columbia Phase 2 trial testing ketamine for Long COVID fatigue and cognitive symptoms (NCT identifier pending, 20 participants)
- Case reports: Published cases demonstrating rapid improvement in treatment-resistant depression and suicidality in Long COVID patients. These reports are focused on psychological impacts. However, there are significant physiological benefits to treatment as well
- Mechanism studies: Ketamine reduces specific inflammatory markers that correlate with Long COVID symptom severity
- Combination therapy: German observational study showed promising results combining ketamine with repetitive transcranial magnetic stimulation (rTMS)
Administration routes:
- Low-Dose Sublingual Ketamine: Although there are other administrative routes (IV, intranasal, etc…), to achieve therapeutic dosing for Long Covid and Post-Vaccine Syndrome patients, only low doses are needed. These doses can be achieved through sublingual administrations via compounded drops, or troches.
Clinical experience: Most dramatic improvements occur in patients with:
- Severe fatigue with cognitive dysfunction
- Chronic neuropathic pain
- Persistent brain fog unresponsive to other interventions
- Anxiety and depressive related disorders
- Dysautonomia symptoms
Typical protocol: Daily sublingual drops or troches at minimally tolerable dose. Dosing dependent upon tolerability is based on glutamate imbalances, which are corrected over time with sublingual low-dose ketamine. Improvements are seen over a 6 month period.
Safety considerations:
- Requires medical supervision and monitoring
- Potential side effects with low-dose sublingual ketamine: Dissociation, minor euphoric feeling, anxiety during infusion
- Contraindications: Active substance abuse (however, this can also be a treatment for substance abuse)
- Not appropriate for all patients
Why adoption is limited: Off-label prescribing of low-dose sublingual ketamine for Long Covid and Post-Vaccine Syndrome is not something many clinicians are aware, let alone comfortable with. Its benefits are well-known in some psychiatric medicine circles for treatment resistant depression, but its effects on BDNF and neuronal healing are not yet mainstream for the treatment of other conditions, such as Long Covid, that impact brain health. It is a controlled substance, which means prescribers must have DEA licenses.
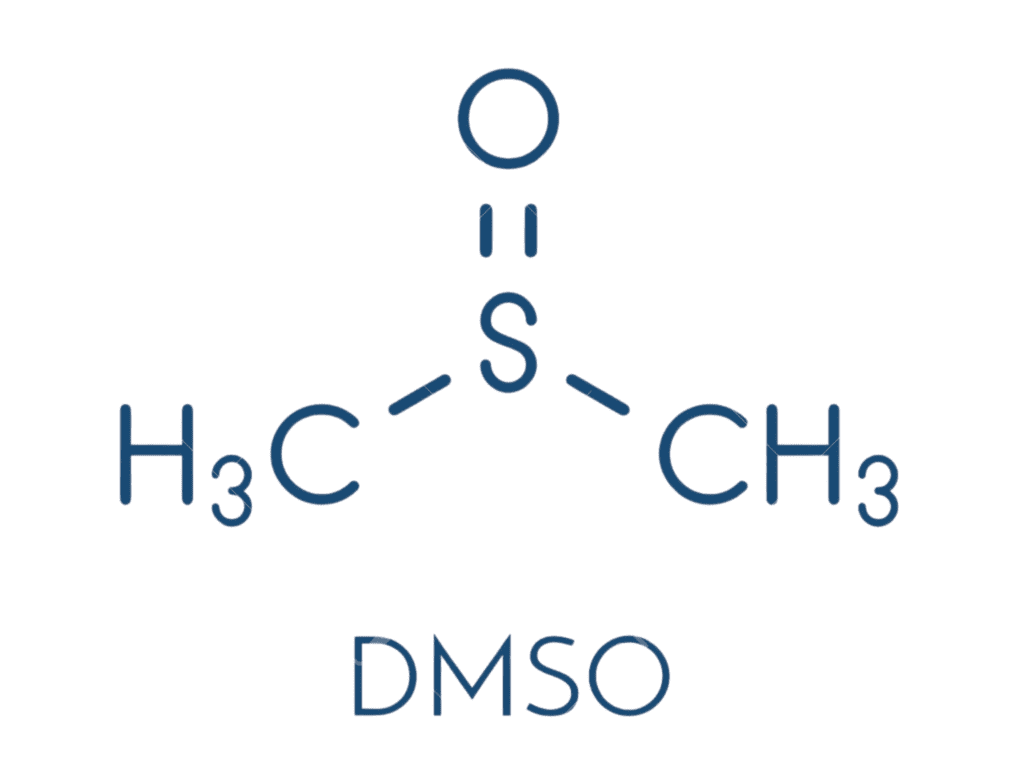
Treatment #3: Dimethyl Sulfoxide (DMSO)
What it is: Dimethyl sulfoxide (DMSO) is a chemical solvent and prescription medication primarily used to treat painful bladder syndrome (interstitial cystitis). It is known for its ability to rapidly penetrate skin and membranes, acting as an anti-inflammatory and analgesic agent.
Clinical rationale for Long COVID and Post-Vaccine Syndrome:
Some argue that DMSO is the most suppressed therapeutic in modern medicine because it works too well for too many things. You’ll remember from earlier in this article what the Goldman Sachs analyst said about effective, curative treatments. When the FDA banned DMSO in 1965 (despite overwhelming safety data), thousands of patients with debilitating conditions lost access to a therapy that was giving them their lives back. The parallels to how Long COVID and Post-Vaccine Syndrome patients are being dismissed today are not coincidental.
Here’s what makes DMSO uniquely suited for spike protein disease:
The Spike-Protein Connection: Why DMSO Makes Mechanistic Sense
If persistent spike protein is driving Long COVID and Post-Vaccine Syndrome pathology—and mounting evidence suggests it is—then DMSO addresses nearly every downstream mechanism we see clinically:
1. Protein Misfolding and Amyloid Fibrin Microclotting
DMSO is a chemical chaperone. It stabilizes protein folding and, critically, dissolves amyloid aggregates.
At least 40 studies have demonstrated DMSO’s ability to solubilize amyloid fibrils and enable the body to break them down. If spike protein is creating misfolded proteins or amyloid-like microclots (the leading hypothesis for many Long COVID and PACVS symptoms), DMSO has a direct mechanism of action:
- Prevents proteins from aggregating into pathologic forms
- Dissolves existing amyloid deposits
- Allows the body to clear these aggregates through normal elimination pathways
This isn’t theoretical, or even in vitro evidence. DMSO has successfully treated amyloidosis in humans—a condition characterized by insoluble protein aggregation causing organ damage. Same mechanism, different protein.
The microclotting angle: DMSO is also a powerful platelet deaggregator and anticoagulant:
- Inhibits platelet aggregation induced by ADP, collagen, arachidonic acid, and platelet-activating factor
- Blocks tissue factor (TF) expression—the key link between inflammation and clotting
- Inhibits thromboxane A2 release
- Increases cAMP (which prevents platelet aggregation)
- Unlike aspirin or heparin, it does this without bleeding risk
If microclots are clogging your microcirculation and causing tissue hypoxia, DMSO addresses it directly while simultaneously improving blood flow to oxygen-starved tissues.
2. Autoimmunity
Spike-triggered autoimmunity is one of the most devastating Long COVID mechanisms. DMSO has decades of evidence treating autoimmune conditions that conventional medicine still can’t touch:
- Lupus – reduces symptoms, often allowing steroid reduction
- Multiple sclerosis – stops progression in many cases, improves symptoms
- Rheumatoid arthritis – 70-80% response rate in multiple studies
- Scleroderma – one of the few things that works for this “untreatable” condition
- Interstitial cystitis – the only FDA-approved use for DMSO (because the evidence was impossible to suppress)
- Sjögren’s syndrome – dramatically improves dry mouth/eyes in published case series
The mechanism appears multifactorial:
- Reduces inflammatory cytokines: IL-1α, IL-1β, IL-6, TNF-α, macrophage chemoattractant protein-1
- Modulates immune cell recruitment and adhesion
- May eliminate pleomorphic bacteria that trigger autoimmunity (a fascinating rabbit hole detailed in the research)
- Reduces immune-mediated tissue damage
If your Long COVID involves autoimmune symptoms—and for many it does—DMSO has evidence where other therapies have failed.
3. Vascular Dysfunction and Endothelial Damage
Spike protein binds to ACE2 receptors on endothelial cells, causing inflammation and vascular dysfunction. DMSO addresses this at multiple levels:
- Vasodilation – relaxes blood vessels, improves perfusion
- Reduces endothelial inflammation – prevents TNF-α induced neutrophil adhesion to vessel walls
- Improves microcirculation – documented in studies using radioactive isotope imaging
- Treats vasospasm – successfully used for Raynaud’s syndrome, peripheral neuropathy, complex regional pain syndrome
Clinical studies show DMSO:
- Eliminates symptoms in 50% of Raynaud’s patients
- Treats diabetic peripheral neuropathy and prevents amputations
- Resolves varicose veins (sometimes within minutes of IV administration)
- Improves circulation in thrombophlebitis and peripheral artery disease
If spike-ACE2 interaction is damaging your vasculature, DMSO protects and heals blood vessels.
4. Neurological Protection and Small Fiber Neuropathy
Small fiber neuropathy is the #4 most common vaccine injury symptom. It’s also prevalent in Long COVID.
DMSO selectively blocks C-fibers and A-delta fibers—the exact nerve fibers responsible for burning pain, electrical shocks, pins-and-needles, and the agonizing dysesthesias of small fiber neuropathy.
Mechanism:
- Blocks nerve conduction in small pain fibers without affecting larger motor/sensory fibers
- Suppresses NMDA and AMPA receptor activity (central pain sensitization)
- Blocks excessive calcium/sodium influx into pain-transmitting neurons
- Does NOT create tolerance (unlike opioids—often becomes MORE effective over time)
Additionally, DMSO crosses the blood-brain barrier and:
- Protects neurons from death following ischemia/hypoxia
- Reduces neuroinflammation
- Treats brain fog, cognitive dysfunction (reported extensively in Long COVID patients using DMSO)
- Has successfully treated traumatic brain injuries, strokes, spinal cord injuries in studies the FDA ignored
5. Organ Protection and Healing
DMSO doesn’t just reduce symptoms, it heals damaged tissue. This is critical for Long COVID and Post-Vaccine Syndrome patients with organ involvement (which would be most patients):
Lungs:
- Reduces pulmonary fibrosis (case reports of transplant candidates recovering)
- Treats COPD, asthma exacerbations
- Improves oxygenation in respiratory insufficiency
- 81% of patients with chronic respiratory failure improved without hospitalization in one study
Gut:
- Heals inflammatory bowel disease (Crohn’s, ulcerative colitis)
- Treats gastritis, peptic ulcers (100% healing rate in refractory cases in one study)
- Normalizes gut permeability (addresses leaky gut)
- Modulates stomach acid production
Heart:
- Protects cardiac tissue from ischemia-reperfusion injury
- Improves cardiac output, reduces vascular resistance
- Successfully used in heart attack patients (before FDA suppression)
Kidneys, liver, pancreas – protective effects documented in dozens of toxicity studies
If Long COVID has damaged your organs, DMSO facilitates healing that conventional medicine cannot.
Clinical Use in Long COVID/PACVS
Who should consider DMSO:
- Patients with microclotting symptoms (exercise intolerance, air hunger, cognitive dysfunction)
- Autoimmune presentations (joint pain, rashes, inflammatory markers)
- Small fiber neuropathy (burning pain, dysesthesias, temperature sensitivity)
- Vascular symptoms (Raynaud’s, livedo reticularis, poor circulation)
- Multi-organ involvement (especially lung, gut, neuro)
Dosing: We start with topical and/or oral dosing based on specific patient needs, titrating appropriately based on response and tolerability. Some patients seek out IV administration for more aggressive treatment, though this is logistically challenging outside specialized clinics.
Expected timeline:
- Acute symptoms (pain, inflammation): Often improve within hours to days
- Chronic conditions: 4-7 days to start seeing benefit, 6-8 weeks for sustained improvement
- DMSO often becomes MORE effective with continued use (opposite of pharmaceutical tolerance)
Contraindications and cautions:
- Pregnancy/breastfeeding – inadequate safety data (though one study showed safe use for infertility)
- Active skin infections – DMSO can carry surface toxins into the body; ensure skin is clean before application
- Severe liver or kidney dysfunction – use with caution, monitor closely
- Concurrent DMSO + PRP injections – may reduce PRP efficacy due to platelet inhibition
- Allergic reactions – rare (~1 in 2000) but possible; start with low concentration to assess tolerance
Common side effects:
- Garlic/oyster-like breath odor (temporary, 2-72 hours)
- Skin irritation with topical use (concentration-dependent, often resolves with continued use)
- Nausea (uncommon with appropriate dosing)
Why You Haven’t Heard About This
The FDA banned DMSO research in 1965 based on a fabricated safety concern (lens opacities in dogs—which never occurred in any other species or in humans). Despite Congressional hearings, outcry from patients and physicians, and a former Secretary of Health and Human Services championing it after using DMSO to treat his wife’s terminal cancer pain, the FDA never relented.
Why? DMSO threatened too many profitable drug markets.
In 1966, over 1,000 researchers presented evidence at the Waldorf Astoria. Zero withdrew their papers despite FDA pressure. The data was overwhelming.
Sixty years later, patients are still suffering from conditions DMSO could treat—because the FDA decided your access to effective medicine was less important than protecting pharmaceutical profits.
For Long COVID patients failed by the medical establishment, DMSO represents what medicine should have been: safe, effective, accessible, and focused on healing rather than management.
Bottom line on DMSO for Long COVID:
If persistent spike protein is causing:
- Protein misfolding → DMSO refolds and stabilizes proteins
- Amyloid microclots → DMSO dissolves aggregates and prevents platelet aggregation
- Autoimmunity → DMSO treats multiple autoimmune conditions
- Vascular dysfunction → DMSO dilates vessels, heals endothelium, improves circulation
- Neurological damage → DMSO protects neurons, blocks pain pathways, crosses BBB
- Organ damage → DMSO protects lungs, gut, heart, kidneys
The mechanism isn’t speculative. The evidence isn’t anecdotal. This is decades of suppressed research finally reaching patients who need it.
Treatment #4: Microcurrent Therapy
What it is: Microcurrent therapy delivers sub-sensory electrical currents in the microampere range — currents so small they mimic the body’s own bioelectrical signals. We commonly have recommended the Arc Microtech device. The Arc device was engineered by Ian Thirkell, a retired English detective who spent years studying the bioelectricity research of Dr. Robert O. Becker after his wife handed him three of Becker’s books and told him to go learn something useful.
The scientific foundation is worth understanding. Becker was an orthopedic surgeon and researcher at the VA Hospital in Syracuse, New York, who spent decades studying how the body uses electrical signals to heal. He discovered that injury generates a measurable shift in voltage at the wound site — a “current of injury” — that initiates the healing process. He demonstrated that applying tiny electrical currents could stimulate partial limb regeneration in rats (published in Nature, 1972) and developed techniques using electrically generated silver ions to fight antibiotic-resistant infections and promote tissue regeneration in human patients. His core insight, detailed in his landmark book The Body Electric (1985): the body operates on a bioelectrical control system that can be supported and restored through the careful application of currents matching the body’s own frequencies. The ARC device is built directly on this principle.
Clinical rationale for Long COVID and Post-Vaccine Syndrome:
ATP production enhancement: The landmark 1982 study by Cheng et al. demonstrated that microcurrent stimulation in the 100-500 microampere range increased ATP production by up to 500% in treated tissue, while increasing amino acid transport by 30-40% and enhancing protein synthesis. When current was increased into the milliampere range used by TENS units, ATP production actually decreased. This distinction matters enormously: Long COVID and Post-Vaccine Syndrome patients have documented mitochondrial dysfunction. A 2025 study in Annals of Medicine confirmed that immune cells from Long COVID patients exhibit aberrant ATP synthase function, resulting in diminished cellular energy availability. The spike protein — whether from viral reservoirs or circulating post-vaccination — impairs oxidative phosphorylation, the process responsible for 90% of cellular energy production. Fatigue, post-exertional malaise, brain fog, exercise intolerance — these are manifestations of cells that cannot produce adequate energy (although there is greater detail as to what is happening here, some of this is detailed in our prior article on cell danger response). Microcurrent directly targets this deficit.
Inflammation reduction: Microcurrent reduces inflammatory markers and promotes resolution of swelling through enhanced circulation and lymphatic drainage — relevant to the chronic systemic inflammation driving Long COVID and Post-Vaccine Syndrome symptoms
Tissue repair and cellular regeneration: Building on Becker’s work, microcurrent stimulates fibroblast activity, enhances collagen synthesis, and promotes angiogenesis. For patients with endothelial damage and microclotting from spike protein, these repair mechanisms are critical. We have seen patients in heart failure restore their ejection fraction back to well over the CHF baseline.
Autonomic and neurological support: Clinical observations from our practice and colleagues in the UK suggest meaningful benefit for patients with autonomic dysfunction, anxiety, depression, and PTSD — conditions that frequently accompany Long COVID and Post-Vaccine Syndrome
One framing we find useful: supplements that support mitochondrial function — CoQ10, B vitamins, magnesium — are static interventions for what is fundamentally a dynamic process. They provide raw materials. Microcurrent is a dynamic intervention: it actively stimulates the cellular machinery to do its job. The two approaches are complementary, not competing.
Current evidence:
Cheng et al. (1982): Foundational study demonstrating 500% ATP increase at therapeutic microcurrent levels, with concurrent increases in amino acid transport and protein synthesis Becker’s published work (1961-1998): Decades of peer-reviewed research on bioelectrical control of regeneration, published in Nature, Science, and other major journals Modern reviews: A 2025 narrative review confirmed microcurrent’s role in enhancing ATP synthesis, improving mitochondrial efficiency, and activating tissue repair mechanisms No randomized controlled trials specific to microcurrent for Long COVID exist, and they may never — putting it in the same category as most treatments on this list when they were first adopted clinically.
Clinical experience: In our practice, over 500 patients have used the ARC device. It has been a reliable and safe recommended interventions for a few years now.
Most consistent improvements:
Fatigue — the most reliably reported benefit, typically noticeable after approximately six weeks of consistent use Pain and inflammation — reduced swelling and bruising, notably effective for patients on anticoagulation therapy Cardiac function — we have observed cases of improved cardiac ejection fraction with sustained use, corroborated by colleagues in the UK Brain fog and cognitive function — improvements likely related to enhanced cellular energy production and reduced neuroinflammation Anxiety and trauma-related symptoms — consistent with observations from military psychiatrists using microcurrent for treatment-resistant PTSD
Benefit is cumulative. More consistent use produces greater results.
Typical protocol:
Program 1 (100% anti-inflammatory): At least one 3-hour cycle daily for two months Program 2 (50% anti-inflammatory, 50% cellular repair): At least one 3-hour cycle daily for two weeks Program 3 (25% anti-inflammatory, 75% cellular repair): At least one 3-hour cycle daily for two weeks Then rotate: One week each of Programs 1, 2, and 3, repeating continuously
Worn on the arm or leg (most patients prefer above the ankle). The strap should be fitted but not tight — two fingers should fit between strap and skin. Tightness inhibits circulation and reduces benefit.
Response timeline: Most patients notice benefit after approximately six weeks. Some report improvement within days, particularly for pain and inflammation. However, some patients even report improved energy in just days.
Safety considerations:
Excellent safety profile — these are sub-sensory currents operating at the same magnitude as the body’s own cellular electrical signaling Approximately 5% of patients experience initial sensitivity (nausea, dizziness), managed by starting with very short sessions and gradually increasing No significant adverse effects observed in our patient population Safe for use alongside other treatments, including anticoagulation Contraindications: Active implanted electrical devices (pacemakers, defibrillators)
Why adoption is limited: Microcurrent therapy sits entirely outside the pharmaceutical paradigm. There is no drug to prescribe, no procedure to bill for, and no pharmaceutical company funding trials. Becker himself faced significant institutional resistance throughout his career — his research challenged the chemical-mechanistic model of biology, and his opposition to electromagnetic pollution put him at odds with powerful interests. The ARC Microtech is a small, family-owned UK company without resources for large-scale clinical trials. And the concept that a wearable device delivering imperceptible electrical currents can meaningfully impact chronic illness strikes most conventionally trained physicians as implausible, despite decades of published research supporting the underlying science.
Our perspective: Microcurrent therapy, and specifically the ARC device, is one of the most underappreciated tools in our clinical arsenal. It is non-invasive, has an excellent safety profile, produces cumulative benefit, and directly addresses what may be the single most important pathological mechanism in Long COVID and Post-Vaccine Syndrome: mitochondrial energy failure. For a condition where the body has lost its ability to produce adequate energy, providing it with the bioelectrical signal to restore that production isn’t alternative quackery — it’s the application of proven biophysics.
Conflicts of Interest: We do offer a discount code for the ARC Microtech device. Using code USARCLEC at checkout on the ARC website provides a $20 discount to ordering customers. ARC also provides us with $20. However, we have never stashed this money away in a bank account. We have historically used this money to re-invest into devices for patients who can not afford to invest in this special device.
Treatment #5: Ivermectin
The controversial treatment that requires honest discussion.
Ivermectin became one of the most politicized medications in modern history. That makes it difficult to have rational, evidence-based conversations about its potential role in Long COVID and Post-Vaccine Syndrome.
Here’s what we actually know:
What it is: An antiparasitic medication on the WHO’s List of Essential Medicines, with an excellent 40-year safety record from billions of doses administered globally.
Theoretical mechanisms for Long COVID and Post-Vaccine Syndrome:
1. Spike Protein and ACE2 Receptor Interaction: This is perhaps the most relevant mechanism for Long COVID and Post-Vaccine Syndrome. Multiple molecular docking studies have demonstrated that ivermectin binds to:
- The spike protein receptor-binding domain (RBD): Where the spike protein attaches to ACE2 receptors
- The spike-ACE2 complex interface: Specifically between leucine 91 of the spike protein and histidine 378 of the ACE2 receptor
- The N-terminal domain (NTD): Which controls initial viral attachment to cell membranes
Binding energy studies show strong affinity (-18 kcal/mol), suggesting ivermectin may physically interfere with spike protein attachment to ACE2 receptors.
Why this matters for Long COVID and Post-Vaccine Syndrome: If persistent spike protein (either from viral reservoirs or circulating spike) is driving symptoms—a leading hypothesis in Long COVID and Post-Vaccine Syndrome pathophysiology—ivermectin’s ability to bind both spike protein and ACE2 receptors could theoretically:
- Block ongoing spike-ACE2 interactions causing cellular dysfunction
- Facilitate clearance of persistent spike protein
- Reduce spike-mediated inflammation and endothelial damage
2. Additional antiviral mechanisms:
- TMPRSS2 binding: Ivermectin binds to TMPRSS2, a protease that activates spike protein for cell entry
- Viral replication inhibition: Demonstrated in vitro inhibition of SARS-CoV-2 replication
- Multiple viral protein targets: Binds to main protease (Mpro), RNA-dependent RNA polymerase (RdRp), and nucleocapsid proteins
3. Anti-inflammatory effects:
- Inhibits nuclear import of inflammatory transcription factors
- Reduces cytokine production (IL-6, TNF-alpha, IL-1β)
- Modulates immune response through multiple pathways
Important concentration caveat: While docking studies predict strong binding, debate exists about whether therapeutically achievable concentrations in humans are sufficient for these effects. One 2024 study suggested effective spike binding required concentrations 100-1000x the approved dosage in their assay system, though other studies and clinical observations suggest benefit at standard doses. Our clinical observations suggest extremely safe dosing ranges are effective in about 70% of patients.
The discrepancy may relate to:
- Differences between in vitro assays and in vivo conditions
- Tissue concentration versus serum concentration
- Individual pharmacokinetic variation
- Duration of exposure in chronic dosing versus single-dose studies
The evidence landscape:
The data on ivermectin for acute COVID-19 was mixed, politicized, and contentious. For Long COVID specifically:
- No published randomized controlled trials
- Mechanistic rationale exists
- Clinical experience varies among practitioners. Our experience suggests a 70% positive clinical response rate
Clinical experience: In our practice, we’ve observed a subset of patients—approximately 70%—who report improvement when ivermectin is included as part of a comprehensive treatment protocol.
Important caveats:
- Not a monotherapy; used as part of multi-modal approach
- Standard anti-parasitic dosing (not megadoses)
- Drug interactions exist
Why adoption is limited: The political controversy surrounding ivermectin creates significant professional risk for physicians who prescribe it, even for legitimate clinical indications. Many state medical boards have issued warnings, and physicians face potential peer censure. Dr. Kory had his board certifications revoked for promoting its use in acutely ill COVID patients, even in spite of positive clinical responses.
Our perspective: With Dr. Kory having testified before the Senate about COVID treatments and faced professional consequences for advocating treatments outside the mainstream, we believe in the efficacy of ivermectin based on excellent safety profile, low cost, and high clinical response rates.
The question for any treatment should be: What does the evidence show, what is the safety profile, and what is the clinical experience?
For ivermectin in Long COVID and Post-Vaccine Syndrome: Mainsteam evidence is limited, the safety profile is excellent, and our clinical experience is positive.
Whether to try it should be an informed decision between patient and physician, weighing potential benefits against minimal risks.
Treatment #6: Anticoagulation Therapy
What it is: Blood-thinning medications ranging from aspirin to low-dose anticoagulants.
Clinical rationale:
Growing evidence suggests microclots may play a role in Long COVID:
- Microclot hypothesis: Persistent microclotting causing tissue hypoxia and symptoms
- Endothelial dysfunction: COVID-19 damages blood vessel lining, promoting clot formation
- Proven benefit in acute COVID: Anticoagulation improves outcomes in hospitalized patients
Current evidence: Mechanistically compelling. Clinical trials underway but results pending.
Clinical experience: Some patients show improvement with aspirin or low-dose anticoagulation. Requires careful patient selection and monitoring.
Safety considerations: Bleeding risk requires careful assessment. Not appropriate for all patients.
Why adoption is limited: Balancing benefit versus bleeding risk requires individualized assessment. Physicians appropriately cautious about anticoagulating without clear indication.
The Gap Between Evidence and Practice
Every treatment discussed here shares common characteristics:
- Mechanistic rationale based on Long COVID and PACVS pathophysiology
- Existing clinical experience supporting use
- Acceptable safety profiles when used appropriately
- Real clinical results within our treatment of over 3,500 Long Covid and Post-Vaccine Syndrome patients
Yet most physicians haven’t adopted them. Why?
The Institutional Reality:
Evidence requirements: Institutions typically require randomized controlled trials before treatment adoption. These trials take years.
Liability concerns: Off-label prescribing, while legal, increases perceived malpractice risk.
Guideline absence: Without official clinical guidelines, physicians lack institutional backing for treatment decisions.
Professional pressure: Medical boards, peer review, and institutional policies create incentives to wait for official recommendations.
This creates a gap: Patients suffering now while waiting for evidence that may take years to generate.
What You Should Know
These Treatments Aren’t Magic Bullets
Long COVID and Post-Vaccine Syndrome are heterogeneous and complex. Treatment response varies significantly between patients.
These treatments should be:
- Part of comprehensive care (not monotherapy)
- Tried systematically (not simultaneously)
- Monitored carefully (medical supervision required)
- Adjusted based on response (iterative approach)
We’re Working With Evolving Evidence
The ideal scenario would be completed randomized controlled trials for each treatment.
What we actually have:
- Mechanistic understanding (how they should work)
- Clinical experience (what we’ve observed)
- Emerging trial data (what’s being formally tested)
- Urgent patient need (can’t wait years for perfect evidence)
The challenge: Balancing the desire for perfect evidence with the reality of patients suffering now.
This Requires Specialized Care
Optimal Long COVID and Post-Vaccine Syndrome treatment requires a clinician who:
- Understands complex post-viral illness
- Can navigate off-label prescribing appropriately
- Will monitor carefully for adverse effects
- Knows when to adjust or discontinue treatment
- Can integrate multiple therapeutic modalities
Not all physicians have this expertise or comfort level.
Clinical Outcomes
In our practice of 3,500+ Long COVID patients:
80-85% achieve significant functional improvement
This means:
- Return to work (often with modifications initially)
- Ability to exercise (frequently at reduced level initially)
- Brain fog resolved or significantly improved
- Fatigue reduced to manageable levels
- Quality of life substantially better
Realistic timeline: 9-18 months on average
Long COVID and Post-Vaccine Sydnrome are chronic conditions requiring sustained treatment. Promises of rapid recovery are unrealistic for most patients.
Finding Appropriate Care
If you’re considering these treatments:
1. Informed discussion with your current physician Share this information. Ask if they’re willing to consider these approaches.
2. Seek Long COVID expertise Find physicians with specific experience treating post-viral illness.
3. Consider telemedicine options Some Long COVID and PACVS specialists work across state lines via telemedicine.
4. Join support communities Other patients can recommend physicians with Long COVID expertise.
5. Be your own advocate Come prepared with questions. Understand the rationale. Participate actively in treatment decisions.
The Bottom Line
Long COVID and PACVS treatment requires:
- Integration of emerging research
- Clinical judgment based on experience
- Willingness to use approved medications for new indications
- Patient-centered approach to risk-benefit analysis
- Honest acknowledgment of uncertainty
Not all physicians are equipped or willing to practice this way. The traditional model of “wait for definitive evidence” serves patients poorly when that evidence is years away.
At our practice, we integrate the best available evidence with clinical experience from over 3,500 cases. We’re transparent about what we know, what we don’t know, and what we’ve observed.
This represents clinical medicine as it should be practiced: Thoughtful, evidence-informed, patient-centered, and unafraid of reasonable uncertainty.
If you’re a Long COVID or Post-Vaccine Syndrome patient seeking care that integrates clinical experience with emerging research, we treat complex post-viral cases at Leading Edge Clinic. Our approach is based on 3,500+ patient encounters, current medical literature, and individualized treatment planning.
Schedule for care: https://drpierrekorypatientportal.md-hq.com/registration
References:
Treatment #1: Low-Dose Naltrexone
Sasso EM, Eaton-Fitch N, Smith P, Muraki K, Marshall-Gradisnik S. Low-dose naltrexone restored TRPM3 ion channel function in natural killer cells from long COVID patients. Frontiers in Molecular Biosciences. 2025;12. doi:10.3389/fmolb.2025.1582967. https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2025.1582967/full
Effect of low dose naltrexone for long covid: a systematic review. medRxiv preprint. 2025. (Meta-analysis: n=155, 4 studies; Hedges’ g = -0.74 for fatigue, SF-36 scores 36.5 to 52.1). https://www.medrxiv.org/content/10.1101/2025.09.09.25335451v1.full
Treatment #2: Low-Dose Ketamine
Baldwin K, Wanson A, Gilecki L-A, Dalton C, Peters E, Halpape K. Intranasal ketamine as a treatment for psychiatric complications of long COVID: a case report. Mental Health Clinician. 2023;13(5):239-243. doi:10.9740/mhc.2023.10.239. PMC10732124. https://pmc.ncbi.nlm.nih.gov/articles/PMC10732124/
Evaluating the Neuromodulatory Effect of Ketamine in Long COVID-19. ClinicalTrials.gov Identifier: NCT06821087. University of British Columbia. https://clinicaltrials.gov/study/NCT06821087
Rolle C, Scheib M, Frank A, Russ I. Treatment of Chronic Fatigue Syndrome (CFS) in Post-SARS-CoV-2 Infection through combined outpatient Neuromodulation Therapy with Repetitive Transcranial Magnetic Stimulation (rTMS) and Ketamine IV Therapy — A Case Series. International Clinical Medical Case Reports Journal. https://ketaminplus.com/en/medical-studies
Zanos P, Moaddel R, Morris PJ, et al. Ketamine and ketamine metabolite pharmacology: insights into therapeutic mechanisms. Pharmacological Reviews. 2018;70(3):621-660. doi:10.1124/pr.117.015198.
Treatment #3: DMSO
Amyloid fibril dissolution / chemical chaperone:
Dzwolak W, Loksztejn A, Smirnovas V. Noncooperative dimethyl sulfoxide-induced dissection of insulin fibrils: toward soluble building blocks of amyloid. Biochemistry. 2009;48(26):6272-6284. https://pubmed.ncbi.nlm.nih.gov/19385641/
Kardos J, Yamamoto K, Hasegawa K, et al. Dissolution of beta2-microglobulin amyloid fibrils by dimethylsulfoxide. Journal of Biological Chemistry. 2003;278(24):21222-21227. https://pubmed.ncbi.nlm.nih.gov/12944383/
Shen CL, Murphy RM. Solvent effects on self-assembly of beta-amyloid peptide. Biophysical Journal. 1995;69(2):640-651. https://pubmed.ncbi.nlm.nih.gov/8527678/
Hoshino M, Katou H, Hagihara Y, et al. Dimethylsulfoxide-quenched hydrogen/deuterium exchange method to study amyloid fibril structure. Biochimica et Biophysica Acta (BBA) – Biomembranes. 2007;1768(8):1886-1899. https://www.sciencedirect.com/science/article/pii/S0005273607000703
Iwai A, Yoshida T, Saito T, et al. Studies on biological actions of dimethyl sulfoxide in familial amyloidosis. Annals of the New York Academy of Sciences. 1983;411:52-64. https://pubmed.ncbi.nlm.nih.gov/6576722/
Amemori S, Iwakiri R, Ootani H, et al. Oral dimethyl sulfoxide for systemic amyloid A amyloidosis complication in chronic inflammatory disease: a retrospective patient chart review. Journal of Gastroenterology. 2006;41:444-449. https://link.springer.com/article/10.1007/s00535-006-1792-3
Platelet aggregation inhibition / anticoagulant properties:
Asmis LM, Bazargan A, Pellegrin M, et al. DMSO inhibits human platelet activation through cyclooxygenase-1 inhibition. A novel agent for drug eluting stents? Biochemical and Biophysical Research Communications. 2010;391(4):1629-1633. https://pubmed.ncbi.nlm.nih.gov/20035720/
Saeed SA, Rasheed H, Ali TH, et al. Effects of dimethyl sulphoxide on aggregation of human blood platelets. Experimental and Molecular Pathology. 1987;46(2):159-169. https://pubmed.ncbi.nlm.nih.gov/2880990/
Rosenblum WI. Dimethyl sulfoxide effects on platelet aggregation and vascular reactivity in pial microcirculation. Annals of the New York Academy of Sciences. 1983;411:110-119. https://pubmed.ncbi.nlm.nih.gov/6410963/
Rosenblum WI. Dimethyl sulfoxide (DMSO) and glycerol, hydroxyl radical scavengers, impair platelet aggregation within and eliminate the accompanying vasodilation of, injured mouse pial arterioles. Stroke. 1982;13(1):35-39. https://www.ahajournals.org/doi/10.1161/01.str.13.1.35
Saeed SA, Connor JD. Differential effects of dimethyl sulfoxide on human platelet aggregation and arachidonic acid metabolism. Biochemical Medicine and Metabolic Biology. 1988;40(2):143-150. https://www.sciencedirect.com/science/article/abs/pii/0885450588901156
Treatment #4: Microcurrent Therapy (ARC Microtech)
Cheng N, Van Hoof H, Bockx E, et al. The effects of electric currents on ATP generation, protein synthesis, and membrane transport in rat skin. Clinical Orthopaedics and Related Research. 1982;(171):264-272.
Becker RO. Stimulation of partial limb regeneration in rats. Nature. 1972;235(5333):109-111.
Becker RO, Spadaro JA. Electrical stimulation of partial limb regeneration in mammals. Bulletin of the New York Academy of Medicine. 1972;48(4):627-641.
Becker RO, Chapin S, Sherry R. Regeneration of the ventricular myocardium in amphibians. Nature. 1974;248(444):145-147.
Becker RO, Selden G. The Body Electric: Electromagnetism and the Foundation of Life. William Morrow & Company; 1985.
Becker RO. Iontopheretic system for stimulation of tissue healing and regeneration. US Patent 5814094A. 1998.
Jonik S, Rothka AJ, Cherin N. Investigating the therapeutic efficacy of microcurrent therapy: a narrative review. Journal of Rehabilitation Medicine. 2025. PMC12357078. https://pmc.ncbi.nlm.nih.gov/articles/PMC12357078/
Piras A, Trofè A, Piperi I, et al. Physiological effects of microcurrent and its application for maximising acute responses and chronic adaptations to exercise. European Journal of Applied Physiology. 2022. https://link.springer.com/article/10.1007/s00421-022-05097-w
Heald AH, Stedman M, Horne L, et al. Mitochondrial function is impaired in long COVID patients. Annals of Medicine. 2025. PMC12344680. https://pmc.ncbi.nlm.nih.gov/articles/PMC12344680/
Hansen KE, et al. Genetic landscape and mitochondrial metabolic dysregulation in patients suffering from severe long COVID. Journal of Medical Virology. 2025. https://onlinelibrary.wiley.com/doi/full/10.1002/jmv.70275
Leppik L, et al. Effects of electrical stimulation on rat limb regeneration, a new look at an old model. Scientific Reports. 2015. PMC4683620. https://pmc.ncbi.nlm.nih.gov/articles/PMC4683620/
Marsland S. Arc Microtech. Lightning Bug (Substack). February 20, 2025. https://lightningbug.substack.com/p/arc-microtech
Treatment #5: Ivermectin
Lehrer S, Rheinstein PH. Ivermectin docks to the SARS-CoV-2 spike receptor-binding domain attached to ACE2. In Vivo. 2020;34(5):3023-3026. PMC7652439. https://pmc.ncbi.nlm.nih.gov/articles/PMC7652439/
Eweas AF, Alhossary AA, Abdel-Moneim AS. Molecular docking reveals ivermectin and remdesivir as potential repurposed drugs against SARS-CoV-2. Frontiers in Microbiology. 2021;11:592908. doi:10.3389/fmicb.2020.592908.
Ahmad S, Waheed Y, Abro A, Abbasi SW, Ismail S. Molecular screening of glycyrrhizin-based inhibitors against ACE2 host receptor of SARS-CoV-2. Structural Chemistry. 2021;32:1441-1452.
[2024 NTD binding study — Ivermectin binds spike N-terminal domain across variants including KP.3. Viruses (MDPI). 2024.]
Treatment #6: Anticoagulation Therapy
Kell DB, Laubscher GJ, Pretorius E. A central role for amyloid fibrin microclots in long COVID/PASC: origins and therapeutic implications. Biochemical Journal. 2022;479(4):537-559. doi:10.1042/BCJ20220016. https://portlandpress.com/biochemj/article/479/4/537/230829/
Pretorius E, Venter C, Laubscher GJ, et al. Prevalence of symptoms, comorbidities, fibrin amyloid microclots and platelet pathology in individuals with Long COVID/Post-Acute Sequelae of COVID-19 (PASC). Cardiovascular Diabetology. 2022;21(1):148. doi:10.1186/s12933-022-01579-5. https://link.springer.com/article/10.1186/s12933-022-01579-5
Pretorius E, Vlok M, Venter C, et al. Persistent clotting protein pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovascular Diabetology. 2021;20:172. doi:10.1186/s12933-021-01359-7.
Grobbelaar LM, Venter C, Vlok M, et al. SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19. Bioscience Reports. 2021;41(8):BSR20210611. doi:10.1042/BSR20210611.
Kruger A, Vlok M, Turner S, et al. Proteomics of fibrin amyloid microclots in long COVID/post-acute sequelae of COVID-19 (PASC) shows many entrapped pro-inflammatory molecules that may also contribute to a failed fibrinolytic system. Cardiovascular Diabetology. 2022;21(1):190. doi:10.1186/s12933-022-01623-4. PMC9491257. https://pmc.ncbi.nlm.nih.gov/articles/PMC9491257/
Libby P, Lüscher T. COVID-19 is, in the end, an endothelial disease. European Heart Journal. 2020;41(32):3038-3044. doi:10.1093/eurheartj/ehaa623.
Medical Disclaimer: This content is for educational purposes only and does not constitute medical advice. All treatments discussed require physician supervision. Consult a qualified healthcare provider before starting any new treatment. Individual results vary. Treatment decisions should be made in consultation with your physician based on your specific medical history and circumstances.