The Buzz Around Benzimidazoles
If you’ve spent any time researching repurposed drugs for cancer, you’ve almost certainly come across fenbendazole. The story of Joe Tippens, a terminal lung cancer patient who attributed his remission to a veterinary deworming drug, caught massive attention in 2019 and hasn’t stopped circulating since. More recently, Mel Gibson’s appearance on The Joe Rogan Experience, where he described friends beating Stage 4 cancers with fenbendazole and ivermectin, pushed the conversation further into the mainstream.
Before the Gibson interview on January 9th 2025, we had been steadily trodding along, a year into our adjunctive cancer program and participation in the Rebuild Medicine observational study. We were already well aware of the power of ivermectin and benzimidazoles.
Cancer patients, especially those with advanced disease who’ve exhausted conventional options, are looking for anything with a reasonable safety profile and a plausible mechanism. And the science behind benzimidazole anthelmintics as anticancer agents is real and growing. We have seen it first-hand, with our patients experience meaningful results, including remissions, through a multi-targeted program using a combination of repurposed drugs – including benzimidazoles. Dr. Kory documented similar successes to Joe Tippens’ story on his substack, detailing 5 patient case studies of Stage 4 Lung Cancer patients.
But here’s what gets lost in the excitement: fenbendazole is not the only benzimidazole, and it may not be the best one for cancer patients. In our clinical practice, we recommend mebendazole over fenbendazole, and not because mebendazole has a better marketing story. The reasons are pharmacological, clinical, and cautionary.
This article covers the benzimidazole drug class, the shared anticancer mechanisms that make them a must-have in our adjunctive cancer program, and why the choice between these drugs matters more than most online discourse acknowledges.
What Are Benzimidazoles?
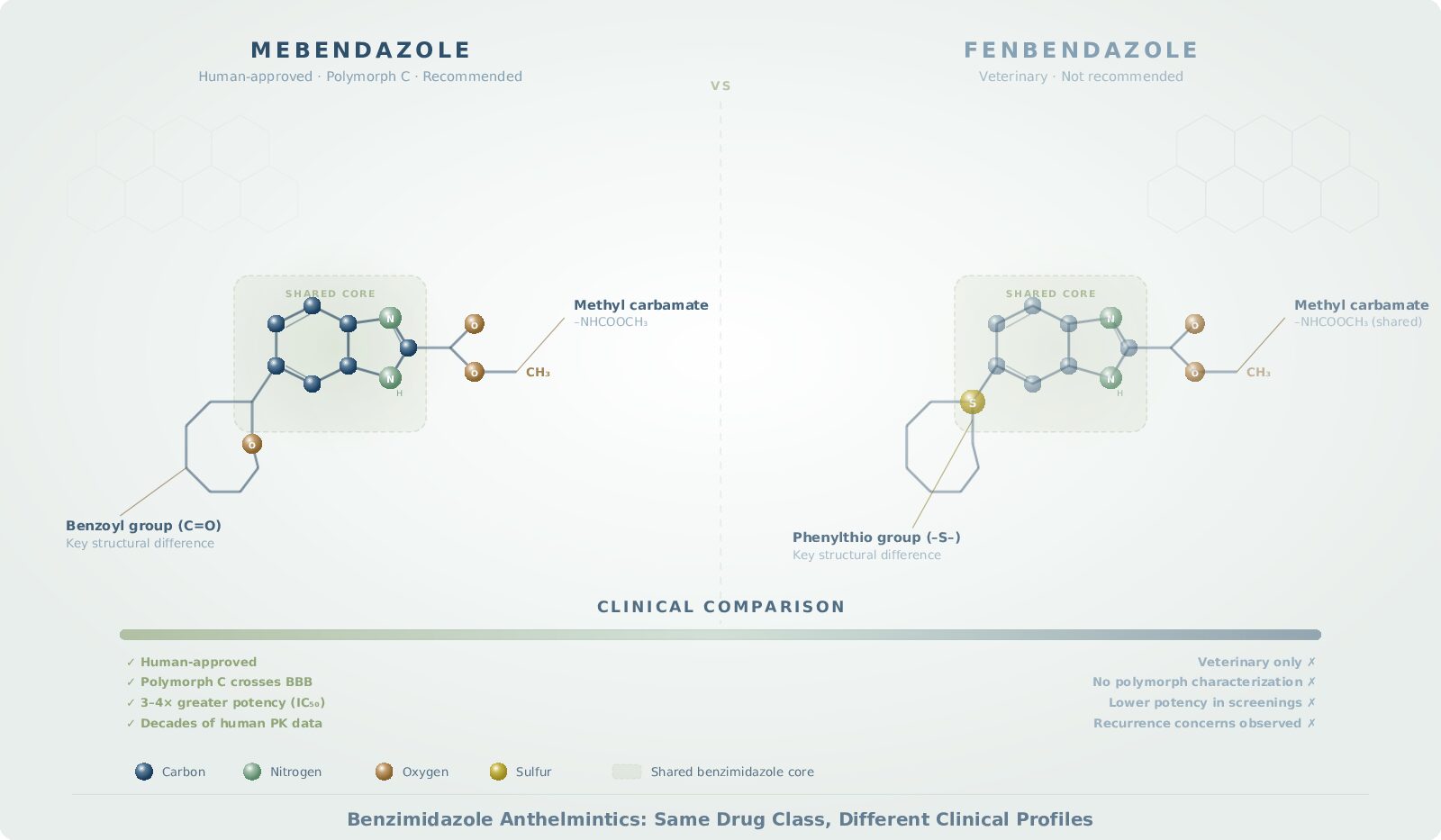
Benzimidazoles are a class of anthelmintic (anti-parasitic) drugs that share a common chemical structure: a benzene ring fused with an imidazole ring. The class includes several drugs relevant to cancer research:
Mebendazole (MBZ): Approved for human use. Used to treat pinworm, roundworm, hookworm, and other parasitic infections. Available from compounding pharmacies.
Fenbendazole (FBZ): Approved for veterinary use. Commonly used in dogs, cats, and livestock. Not approved for human use by the FDA.
Albendazole (ABZ): Approved for human use. Primarily used for echinococcosis and neurocysticercosis. Less studied for cancer applications than mebendazole.
All three share the same core mechanism of action—they bind to β-tubulin and disrupt microtubule formation. This is why their anticancer properties overlap significantly. But they are not identical drugs. Their pharmacokinetics, bioavailability, polymorphic forms, regulatory status, and clinical track records in cancer patients differ in ways that have real consequences for patient outcomes.
How Benzimidazoles Fight Cancer: Shared Mechanisms
The anticancer activity of benzimidazoles has been demonstrated across dozens of preclinical studies and is now supported by an emerging clinical evidence base. Dr. Paul Marik classifies the mebendazole/fenbendazole/albendazole group as Tier One repurposed drugs in his comprehensive cancer care monograph (Marik, 2024)—meaning there is strong recommendation based on the available evidence.
The mechanisms are multi-targeted and address several cancer hallmarks simultaneously.
1. Microtubule Disruption and Mitotic Arrest
This is the foundational mechanism and the one benzimidazoles were essentially designed for, albeit against parasites, not cancer.
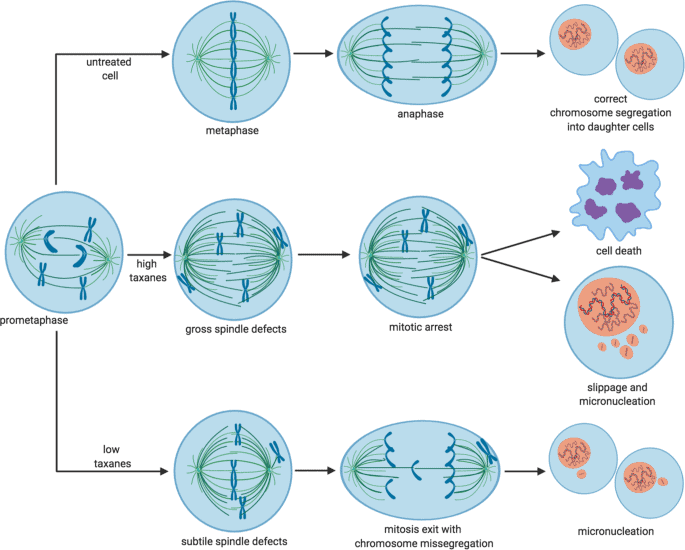
Microtubules are structural proteins that form the “skeleton” of dividing cells. During cell division (mitosis), microtubules organize into the mitotic spindle, which pulls chromosomes apart so each daughter cell gets a complete copy of DNA. Cancer cells divide rapidly and depend heavily on functional microtubules to do so.
Benzimidazoles bind to β-tubulin and prevent it from polymerizing into microtubules. Without functional microtubules, the mitotic spindle cannot form, and cell division arrests. Cells stuck in mitotic arrest eventually undergo apoptosis (programmed cell death).
This is the same general mechanism used by taxane chemotherapy drugs (paclitaxel, docetaxel) and vinca alkaloids (vincristine, vinblastine), except benzimidazoles achieve it at a fraction of the toxicity and cost.
In comparative screenings, mebendazole has demonstrated the greatest inhibitory effect among benzimidazoles tested against cancer cell lines, with IC50 values of 0.30–0.32 µM in chemoresistant melanoma cells—compared to 0.7–1.2 µM for albendazole and 1.2–1.4 µM for fenbendazole (Doudican et al., 2008). Importantly, mebendazole showed significantly less toxicity to normal melanocytes, demonstrating selectivity for cancer cells.
2. p53 Reactivation and Apoptosis Induction
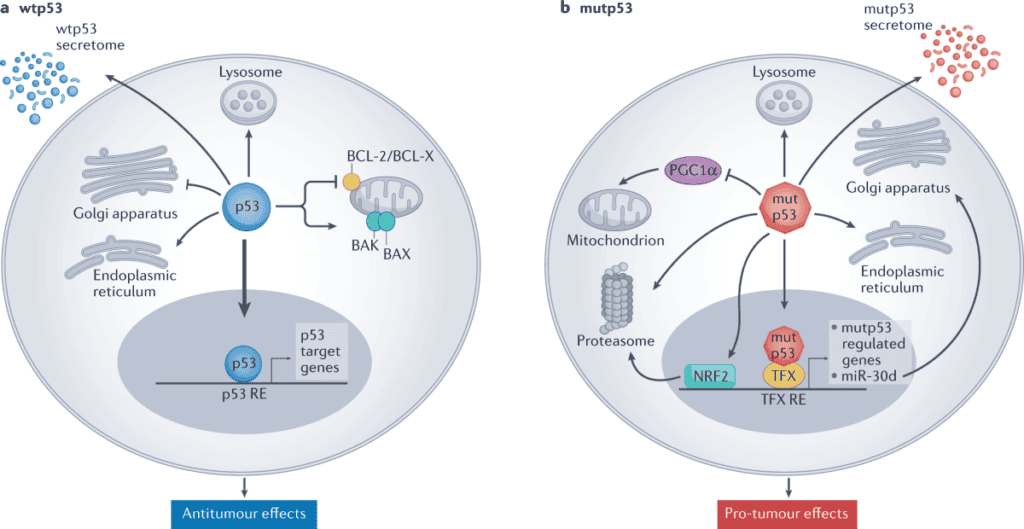
The p53 tumor suppressor gene, often called the “guardian of the genome”, is the most commonly mutated gene in human cancers. When functional, p53 detects DNA damage and either halts cell division for repair or triggers apoptosis if the damage is irreparable. In many cancers, p53 is suppressed or rendered non-functional by overexpression of its negative regulators, MDM2 and MDMX.
Benzimidazoles have been shown to downregulate both MDM2 and MDMX, effectively restoring p53 function in tumor cells where p53 itself remains intact but is being suppressed (Mrkvová et al., 2019). This reactivation of p53 shifts the balance from uncontrolled proliferation back toward programmed cell death.
The upregulation of p53 and its downstream target p21 has been demonstrated for both mebendazole and fenbendazole (Dogra et al., 2018; Doudican et al., 2008), confirming this as a class-level effect.
3. Metabolic Disruption: Targeting the Warburg Effect
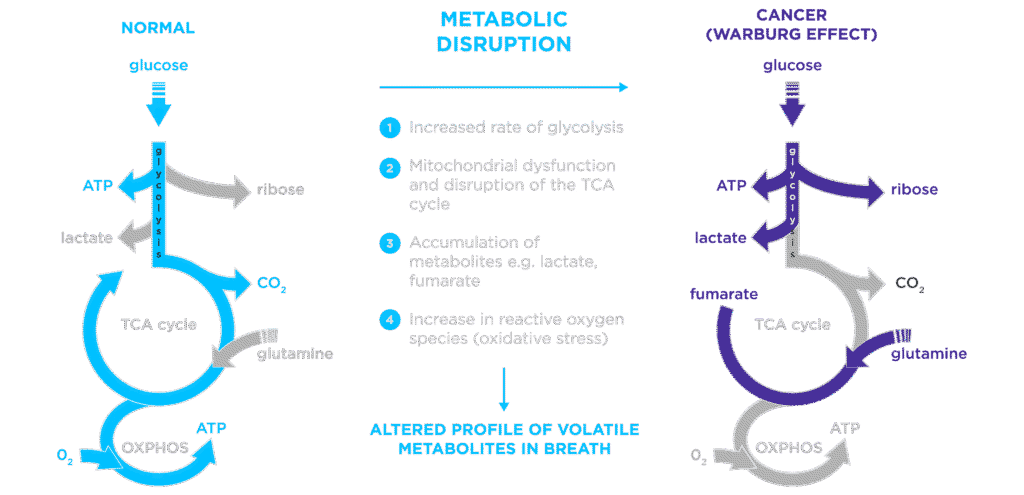
Cancer cells are metabolic parasites (not literal parasites, though). Rather than relying on efficient mitochondrial oxidative phosphorylation like healthy cells, most cancer cells depend heavily on glycolysis, fermenting glucose even in the presence of oxygen. This metabolic reprogramming, known as the Warburg effect, is now recognized as a fundamental hallmark of cancer and the basis for the metabolic theory of cancer as articulated by Thomas Seyfried and others.
Benzimidazoles inhibit glucose uptake and glycolytic enzymes in cancer cells, starving them of their primary fuel source (Pantziarka et al., 2014). This metabolic disruption is particularly relevant in the context of a ketogenic dietary intervention, a strategy we employ – when indicated – alongside repurposed drug protocols at our practice. When you restrict glucose availability through diet and simultaneously block cancer cells’ ability to import what glucose remains, you create a hostile metabolic environment that healthy cells can adapt to, but cancer cells cannot.
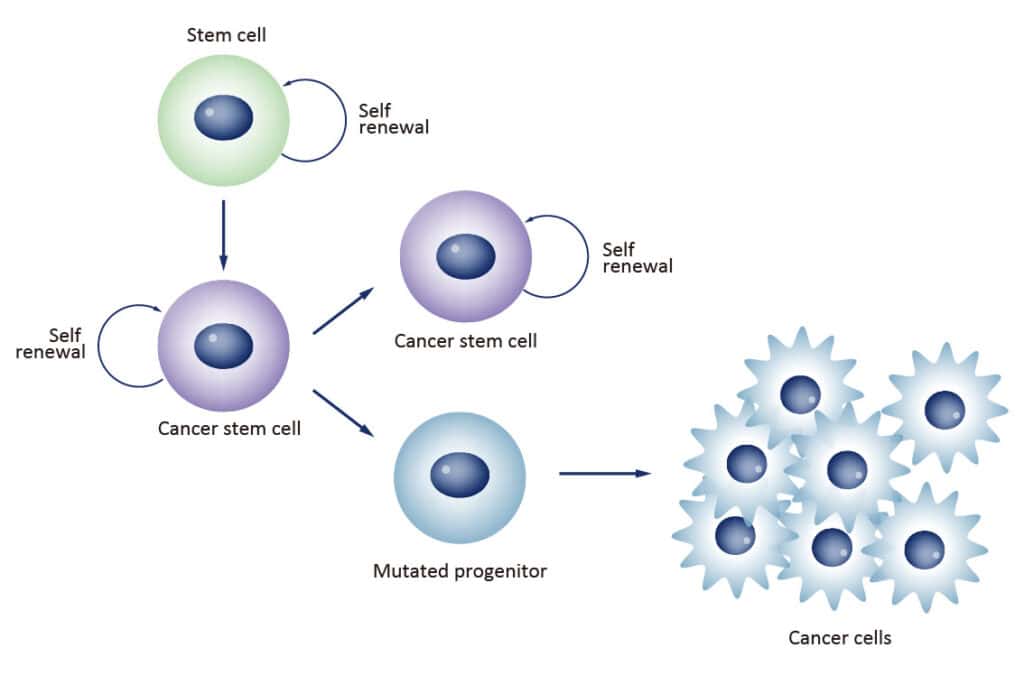
4. Cancer Stem Cell Targeting
Cancer stem cells (CSCs) are the subpopulation of cells within a tumor responsible for treatment resistance, recurrence, and metastasis. They are the reason cancers come back after chemotherapy appears to have worked. Conventional cytotoxic treatment kills the bulk tumor cells while leaving the CSCs intact.
This is one of the central differentiators of our adjunctive cancer care program: we target cancer stem cells directly, while conventional treatment largely does not.
Benzimidazoles target CSCs through multiple pathways:
Sonic Hedgehog/GLI1 inhibition: Mebendazole inhibits the Hedgehog signaling pathway—a major driver of cancer stem cell self-renewal—at IC50 of 0.516 µM, positioning it as a potential alternative to the pharmaceutical Hedgehog inhibitor vismodegib (Borodovsky et al., 2015).
ALDH+ stem cell depletion: Mebendazole depletes ALDH+ cancer stem cells in triple-negative breast cancer, one of the most aggressive and difficult-to-treat subtypes (Targeting CSCs, as reviewed in Marik, 2024).
Wnt/β-catenin pathway suppression: Both mebendazole and fenbendazole interfere with Wnt signaling, which is critical for CSC maintenance and self-renewal across multiple cancer types.
When combined with ivermectin, which targets CSCs through complementary mechanisms (PAK1/Akt/mTOR blockade, downregulation of NANOG/SOX2/OCT4), the benzimidazole-ivermectin combination provides multi-pathway CSC coverage. This is why we typically recommend both drugs together as part of a comprehensive protocol. Furthermore, we do not recommend these drugs as monotherapies.
5. Anti-Angiogenic Effects
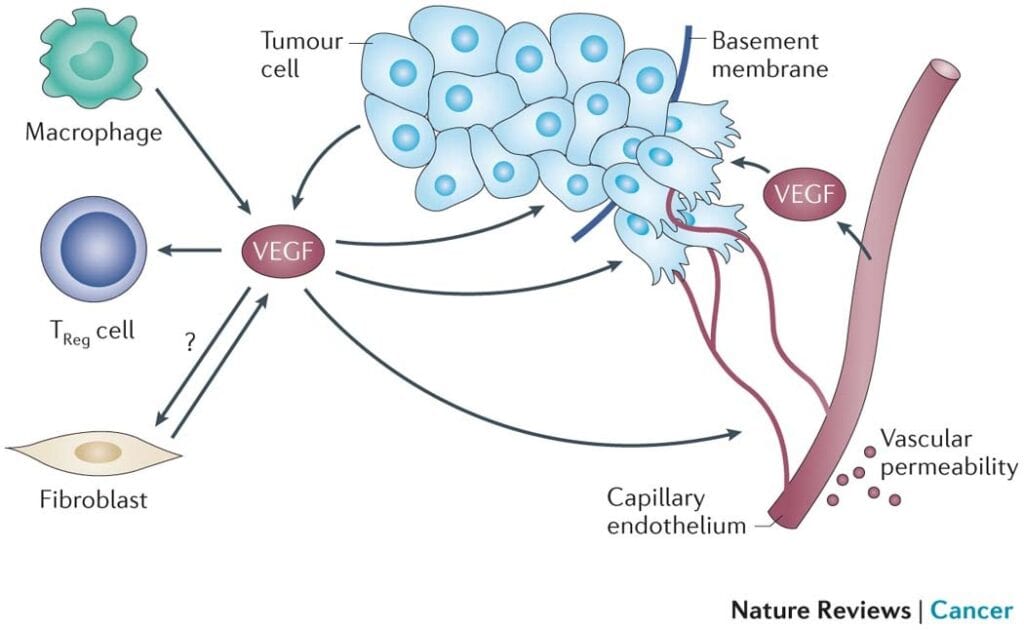
Tumors require a blood supply to grow. The process of recruiting new blood vessels, angiogenesis, is driven largely by vascular endothelial growth factor (VEGF) signaling. Mebendazole reduces VEGFR2 kinase activity and decreases tumor microvessel density in preclinical models, effectively cutting off the tumor’s supply lines (Pantziarka et al., 2014).
This anti-angiogenic property complements the metabolic disruption described above: you’re simultaneously restricting the tumor’s fuel delivery (angiogenesis inhibition) and its ability to use that fuel (glycolysis inhibition).
6. Immune Modulation
Emerging research demonstrates that mebendazole polarizes tumor-associated macrophages from the immunosuppressive M2 phenotype toward the pro-inflammatory M1 phenotype (as reviewed in Marik, 2024). In simple terms: tumors actively suppress local immune cells to avoid detection. Mebendazole helps reverse that suppression, re-enabling the immune system to recognize and attack cancer cells.
This immune-modulating effect is particularly interesting in the context of combination protocols, as it may enhance the activity of other immunomodulatory agents in the regimen, including low-dose naltrexone, which several of our cancer patients also use.
7. BRAF/MEK Pathway Inhibition
Mebendazole inhibits the BRAF/MEK signaling pathway, including the BRAF V600E mutation, one of the most common oncogenic mutations, found in approximately 50% of melanomas and a significant percentage of colorectal, thyroid, and lung cancers. This gives mebendazole a targeted therapy-like mechanism in addition to its broader anticancer effects.
Blood-Brain Barrier Penetration: A Critical Advantage for Mebendazole
One of mebendazole’s most clinically significant properties is its ability to cross the blood-brain barrier (BBB).
Bai et al. (2015) demonstrated in preclinical glioblastoma models that mebendazole achieves therapeutically effective concentrations in brain tissue and tumor. However – and this is critical – not all forms of mebendazole are equal in this regard.
Mebendazole exists in three polymorphic forms: A, B, and C. These are chemically identical but differ in their crystal structure, which affects how they dissolve, absorb, and distribute in the body.
Polymorph C reaches therapeutic concentrations in brain tissue with fewer side effects than polymorphs A or B (Bai et al., 2015). In the preclinical glioblastoma study, polymorph C significantly extended survival, and its efficacy was further enhanced when combined with elacridar (a drug that inhibits the P-glycoprotein efflux pump at the BBB).
Polymorph A showed inferior brain penetration and reduced anticancer efficacy compared to polymorph C.
Polymorph B is the least bioavailable form and is generally considered subtherapeutic for cancer applications.
This distinction matters enormously for patients, and it’s one that most online fenbendazole resources don’t address because fenbendazole doesn’t have this level of polymorphic characterization in human pharmacokinetic studies. If you’re taking mebendazole for cancer and you’re not receiving polymorph C, you may not be getting therapeutic benefit. This is why we recommend obtaining mebendazole from compounding pharmacy partners who can verify polymorph C formulation through 3rd party lab testing, rather than purchasing international products where the polymorphic form is uncertain. This is also why we work with trusted compounding pharmacy partners.
Why We Recommend Mebendazole Over Fenbendazole
Given that benzimidazoles share a core mechanism of action, why does the specific drug choice matter? Several reasons:
1. Human Pharmacokinetic Data
Mebendazole has decades of human pharmacokinetic data from its approved use treating parasitic infections, including long-term, high-dose studies in echinococcosis patients (40–50 mg/kg/day for months to years). This gives clinicians a well-characterized safety profile with known drug interactions, metabolism pathways, and adverse effect patterns.
Fenbendazole has pharmacokinetic data primarily in veterinary species. While human case reports exist, the pharmacokinetic profile in humans is less well-defined. Dosing guidance for fenbendazole in cancer patients is largely extrapolated from veterinary data and anecdotal reports, which provides a less solid foundation for clinical decision-making. This isn’t to say fenbendazole is not safe. If you look at online discussions, many patients self administer fenbendazole without issue. However, this is not something we recommend.
2. Superior Preclinical Potency
In direct comparisons across cancer cell lines, mebendazole has consistently demonstrated greater anticancer potency than fenbendazole. In the Doudican et al. (2008) melanoma screening, mebendazole achieved IC50 values 3-4x lower than fenbendazole (0.30–0.32 µM vs. 1.2–1.4 µM). Nygren et al. (2013), screening 1,600 existing drugs against colon cancer cell lines, identified mebendazole as a top candidate from among a cluster of benzimidazoles that also included fenbendazole.
3. BBB Penetration with Characterized Polymorphism
As detailed earlier, mebendazole polymorph C has published data demonstrating brain tissue penetration at therapeutic concentrations. For patients with primary brain tumors or brain metastases, which represent a significant portion of advanced cancer cases, this is not a theoretical advantage. It is a practical one that can influence treatment outcomes.
4. Clinical Observations on Recurrence Patterns
This is where we share something that we believe patients need to hear, with the caveat that these are clinical observations, rather than published research.
Our compounding pharmacy colleagues, who have worked in the repurposed drug cancer space for nearly two decades, have tracked a concerning pattern among fenbendazole users. Many patients who took fenbendazole had excellent clinical results while on the drug. However, when cancer recurred after discontinuation, it tended to be more aggressive and significantly more resistant to treatment than would typically be expected.
We want to be transparent: this is observational data, not research. But it comes from nearly 18 years of clinical observation across a large number of patients, and we take it seriously. The pattern is consistent with what we know about cancer’s ability to adapt to selective pressures. If fenbendazole exerts a different pharmacodynamic profile than mebendazole, even subtly, it could create different resistance patterns upon discontinuation.
This observation reinforces two principles we hold firmly in our practice:
First, drug cycling matters. Cancer adapts. Any single-agent approach, no matter how effective initially, creates selective pressure that favors resistant cell populations. We rotate and cycle our protocols specifically to prevent this.
Second, these are not monotherapies. No single repurposed drug should be used in isolation. The multi-pathway, multi-drug approach is not just theoretically superior, it is clinically essential for durable outcomes.
5. Regulatory and Quality Control Considerations
Mebendazole is a human-approved pharmaceutical. When obtained from a reputable compounding pharmacy that verifies polymorph C, patients can have confidence in what they are receiving.
Fenbendazole is a veterinary product. The formulations available to consumers are manufactured to veterinary standards, which differ from pharmaceutical-grade human medications. Purity, consistency, and excipient profiles may vary. Patients self-sourcing fenbendazole from pet supply stores or online vendors have no way to verify what they’re actually getting.
The Evidence Base: Cancer Types Studied
Benzimidazoles have demonstrated anticancer activity across an extensive range of cancer types in preclinical studies:
Glioblastoma/Brain cancers: Mebendazole significantly extended survival in preclinical glioblastoma models and crosses the blood-brain barrier (Bai et al., 2011; Bai et al., 2015). Combination with temozolomide (standard chemotherapy) extended survival further than temozolomide alone.
Melanoma: Mebendazole identified as a lead compound from screening 2,000 molecules against chemoresistant melanoma cell lines. Induced dose-dependent apoptosis with selectivity for cancer cells over normal melanocytes (Doudican et al., 2008).
Colorectal cancer: Both mebendazole and fenbendazole identified in drug screens against colon cancer cell lines. Fenbendazole additionally showed activity against 5-fluorouracil-resistant colorectal cancer cells, relevant for patients whose cancers have stopped responding to standard chemotherapy (Nygren et al., 2013; Park et al., 2019).
Breast cancer: Mebendazole and albendazole caused selective apoptotic cell death in breast cancer cell lines while sparing normal cells (Petersen & Baird, 2021). Mebendazole depletes ALDH+ cancer stem cells in triple-negative breast cancer.
Lung cancer: Benzimidazoles have been investigated for both non-small cell lung cancer and lung adenocarcinoma. In our own case series of five metastatic lung cancer patients, polymorph C mebendazole was part of every patient’s treatment protocol alongside ivermectin and other repurposed agents. Outcomes ranged from complete remission (in an ALK-positive NSCLC patient) to sustained disease stability over 6–16 months, including in patients in their 80s and one patient receiving no conventional treatment at all.
Pancreatic cancer: Emerging evidence supports benzimidazole activity in pancreatic cancer models, particularly when combined with metabolic interventions.
Ovarian, gastric, and osteosarcoma: Additional preclinical data across these cancer types further supports the broad-spectrum anticancer activity of the benzimidazole class.
Synergy with Ivermectin: Why We Recommend Both
Mebendazole and ivermectin target cancer through different but complementary mechanisms. The first peer-reviewed protocol for the combined use of ivermectin, mebendazole, and fenbendazole in cancer was published in 2024 (Baghli, Martinez, Marik et al., 2024), providing a formal evidence-based framework for this combination approach.
The rationale for combining them:
Different primary mechanisms: Mebendazole disrupts microtubules; ivermectin disrupts PAK1/Akt/mTOR signaling, mitochondrial function, and WNT pathways. Together, they cover more cancer hallmarks simultaneously.
Complementary CSC targeting: Ivermectin targets cancer stem cells through stemness gene downregulation (NANOG, SOX2, OCT4). Mebendazole targets CSCs through Hedgehog/GLI1 inhibition and ALDH+ depletion. Different mechanisms, same critical target.
Reduced resistance potential: Multi-drug combinations reduce the likelihood that cancer cells can develop resistance to all agents simultaneously. This is the same principle behind multi-drug chemotherapy regimens. But, applied to safer, more tolerable medications.
Published synergistic effects: The combination has shown additive or synergistic anticancer effects in published preclinical data. In our clinical practice, every patient in our metastatic lung cancer case series received both polymorph C mebendazole and ivermectin as part of their multi-drug protocol—and the outcomes in that cohort, including a complete remission and multiple cases of sustained disease stability, are consistent with the synergistic benefit the research predicts.
For a detailed discussion of ivermectin’s independent anticancer mechanisms and our clinical outcomes data, see our companion article: Ivermectin for Cancer: Scientific Mechanisms & Clinical Outcomes.
Clinical Considerations
Administration
Mebendazole should be taken with a fatty meal to improve absorption, as it has relatively low oral bioavailability (~17–20% of the dose reaching systemic circulation). Fat-soluble drugs require dietary fat for adequate absorption. This is not optional, it meaningfully affects therapeutic drug levels.
Specific dosing, cycling schedules, and protocol details are individualized in our practice based on cancer type, stage, concurrent treatments, and patient tolerability. We strongly recommend against self-dosing based on internet protocols. The difference between a thoughtful, clinician-supervised multi-drug regimen with appropriate cycling and a self-administered single-agent protocol is the difference between informed treatment and a gamble.
Safety Profile
Mebendazole has a well-established safety record from decades of human use, including long-term high-dose regimens for echinococcosis. The WHO includes mebendazole on its List of Essential Medicines.
Safety was formally evaluated across 6,276 subjects in 39 clinical trials plus decades of post-marketing experience (Guerini et al., 2019). The most common adverse effects at standard dosing are mild gastrointestinal symptoms. At higher doses used for echinococcosis, hepatotoxicity is the primary concern requiring monitoring, but this is typically at doses significantly above those used in most cancer protocols.
Drug interactions exist and must be managed, particularly with:
- Medications metabolized by CYP enzymes
- Other hepatotoxic agents
- Concurrent chemotherapy (which requires careful coordination)
Contraindications
- Pregnancy (teratogenic in animal studies)
- Significant hepatic impairment (requires monitoring of liver function)
- Known hypersensitivity to benzimidazoles
Monitoring
Patients on mebendazole as part of a cancer protocol should have regular liver function monitoring, complete blood counts, and imaging per their oncologist’s schedule. This is standard practice at Leading Edge Clinic. Our nursing team provides proactive check-ins throughout treatment to manage dosing, side effects, and protocol adherence.
What This Means for Cancer Patients
If you’ve found your way to this article because you’re researching fenbendazole for cancer, here’s what we want you to take away:
The science behind benzimidazoles as anticancer agents is real. The mechanisms are well-characterized, the preclinical data is extensive, and the emerging clinical evidence is encouraging. Naysayers call this wishful thinking, placebo effect, and quackery. But, it is proven pharmacology.
Mebendazole is the superior clinical choice within this drug class for human cancer patients. This statement is based on potency data, human pharmacokinetic characterization, polymorph-specific brain penetration research, and 18 years of clinical observations from compounding pharmacy colleagues.
No benzimidazole should be used as a monotherapy. Cancer adapts. Multi-pathway coverage – using benzimidazoles alongside ivermectin, metabolic interventions, and other repurposed agents – provides the best chance at durable outcomes.
Cycling and protocol management matter. The same adaptability that makes cancer deadly also means static, unchanging drug regimens lose effectiveness over time. We cycle our protocols specifically to stay ahead of resistance.
Clinical supervision is essential. Drug interactions, polymorph verification, dosing optimization, monitoring, and integration with conventional treatment all require experienced clinical management. Our adjunctive cancer care program provides exactly this—frequent provider visits, dedicated nursing support, and protocols built on the research of Dr. Paul Marik and our own clinical experience.
If you’re a cancer patient interested in incorporating mebendazole and other repurposed drugs into your care, learn more about our adjunctive cancer care program or register to make an appointment.
References
Baghli I, Martinez P, Marik PE, et al. Ivermectin, Fenbendazole and Mebendazole protocol in cancer. Published September 19, 2024. Peer-reviewed protocol for combined benzimidazole use in cancer.
Bai RY, Staedtke V, Aprhys CM, Gallia GL, Riggins GJ. Antiparasitic mebendazole shows survival benefit in 2 preclinical models of glioblastoma multiforme. Neuro-Oncology. 2011;13(9):974-982. doi:10.1093/neuonc/nor077
Bai RY, Staedtke V, Wanjiku T, Bhatt R, Riggins GJ. Brain penetration and efficacy of different mebendazole polymorphs in a mouse brain tumor model. Clinical Cancer Research. 2015;21(15):3462-3470. doi:10.1158/1078-0432.CCR-14-2681
Borodovsky A, Larsen AR, Bai RY, et al. Repurposing the antihelmintic mebendazole as a Hedgehog inhibitor. Molecular Cancer Therapeutics. 2015;14(1):3-13. doi:10.1158/1535-7163.MCT-14-0755-T
Dogra N, Kumar A, Mukhopadhyay T. Fenbendazole acts as a moderate microtubule destabilizing agent and causes cancer cell death by modulating multiple cellular pathways. Scientific Reports. 2018;8:11926. doi:10.1038/s41598-018-30158-6
Doudican NA, Byron SA, Pollock PM, Orlow SJ. XIAP downregulation accompanies mebendazole growth inhibition of melanoma xenografts. Anti-Cancer Drugs. 2008;19(2):157-163. doi:10.1097/CAD.0b013e3282f44b67
Guerini AE, Triggiani L, Maddalo M, et al. Mebendazole as a candidate for drug repurposing in oncology: an extensive review of current literature. Cancers. 2019;11(9):1284. doi:10.3390/cancers11091284. PMC6769799. https://pmc.ncbi.nlm.nih.gov/articles/PMC6769799/
Marik PE. Cancer Care: Repurposed Drugs & Metabolic Interventions in Treating Cancer. 2nd ed. Independent Medical Alliance; 2024. https://imahealth.org/wp-content/uploads/2023/06/Cancer-Care-FLCCC-Dr-Paul-Marik-v2.pdf
Mrkvová Z, Uldrijan S, Pombinho A, Bartůněk P, Slaninová I. Benzimidazoles downregulate Mdm2 and MdmX and activate p53 in MdmX overexpressing tumor cells. Molecules. 2019;24(11):2152. doi:10.3390/molecules24112152
Nygren P, Fryknäs M, Agerup B, Larsson R. Repositioning of the anthelmintic drug mebendazole for the treatment for colon cancer. Journal of Cancer Research and Clinical Oncology. 2013;139(12):2133-2140. doi:10.1007/s00432-013-1539-5
Pantziarka P, Bouche G, Meheus L, Sukhatme V, Sukhatme VP. Repurposing drugs in oncology (ReDO)—mebendazole as an anti-cancer agent. ecancermedicalscience. 2014;8:443. doi:10.3332/ecancer.2014.443. https://ecancer.org/en/journal/article/443-repurposing-drugs-in-oncology-redo-mebendazole-as-an-anti-cancer-agent
Park D, Lee JH, Yoon SP. Anti-cancer effects of fenbendazole on 5-fluorouracil-resistant colorectal cancer cells. Korean Journal of Physiology & Pharmacology. 2019;23(5):377-386. doi:10.4196/kjpp.2019.23.5.377
Petersen JSSM, Baird SK. Treatment of breast and colon cancer cell lines with anti-helmintic benzimidazoles mebendazole or albendazole results in selective apoptotic cell death. Journal of Cancer Research and Clinical Oncology. 2021;147(10):2945-2953. doi:10.1007/s00432-021-03698-0
Disclaimer: This article is for educational purposes only and should not be construed as medical advice. Cancer treatment decisions should be made in consultation with qualified oncology professionals. The information presented represents current research and clinical observations; outcomes may vary. Patients should never discontinue or modify standard cancer treatments without consulting their healthcare team.