Introduction: The Clotting Problem Most Doctors Aren’t Testing For
Many Long COVID and Post-Vaccine Syndrome patients know their symptoms. Fatigue that doesn’t resolve with rest. Brain fog that feels like you’re wading through muck just to formulate a coherent thought. Breathlessness that appears without warning. Chest tightness that comes and goes. Post-exertional crashes that wipe out any attempt at normal activity.
What most patients, and most clinicians, have come to know is that for a significant proportion of these patients, the blood itself is part of the problem.
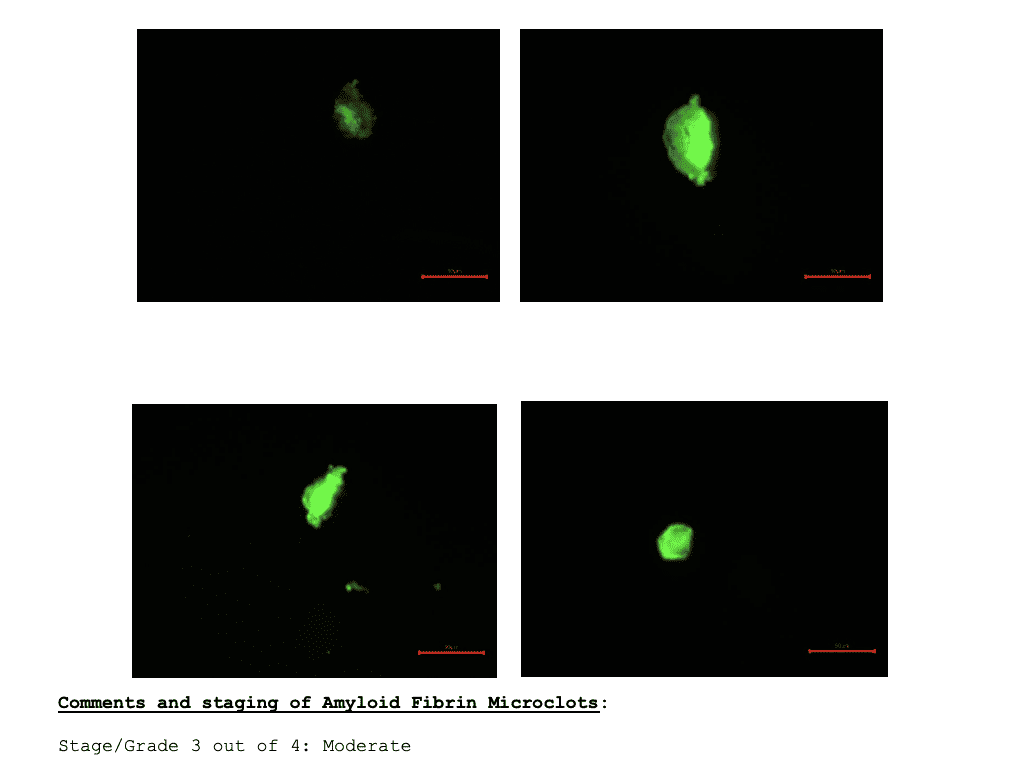
Not in the dramatic, visible way that shows up on imaging. In a quieter, more insidious way: tiny, abnormal clots forming throughout the microvasculature, reducing oxygen delivery to tissues, trapping inflammatory molecules, and physically obstructing circulation in capillaries too small to show up on any standard scan. Amyloid fibrin microclots are not a novel issue. But, with spike protein, they are more significant than ever before. Not a single patient of ours comes to us with low levels of microclotting.
A critical aspect of microclotting we will explore in this article: a genetic variant that approximately 25–30% of the general population carries means some patients are dramatically more prone to forming these clots, and dramatically less able to clear them, than others.
Understanding this mechanism, and how your genetics interact with it, changes what treatment should look like. At Leading Edge Clinic, it’s something we assess in patients where microclotting is suspected as a significant driver. This post explains the underlying science and the clinical implications.
What Are Fibrin Amyloid Microclots?
Normal blood clotting is a tightly regulated process. When a vessel is injured, a soluble protein circulating in plasma called fibrinogen is converted to fibrin, forming a mesh-like clot that stops bleeding. Once the injury heals, an enzyme called plasmin dissolves the fibrin through a process called fibrinolysis. The clot clears. Normal circulation resumes.
In Long COVID and Post-Vaccine Syndrome, this process goes wrong in a specific and well-documented way.
Research pioneered by Professor Etheresia Pretorius at Stellenbosch University and Professor Douglas Kell at the University of Liverpool, beginning in 2021 and now confirmed across multiple independent research groups, has established that the spike protein of SARS-CoV-2 can trigger fibrinogen to misfold into an abnormal, amyloid-like form. These fibrinaloid microclots, the term used in the published literature, have structural properties that make them fundamentally different from normal clots.
Most critically: they resist normal fibrinolysis. The body’s standard clot-clearing machinery, plasmin, cannot effectively break them down.
What Makes These Microclots Different
Normal fibrin clots form a loose mesh that plasmin can penetrate and degrade. Amyloid fibrin microclots are densely compacted, beta-sheet rich structures. This is the same structural architecture seen in amyloid proteins associated with Alzheimer’s and Parkinson’s diseases. Plasmin can penetrate normal clots. It cannot efficiently dissolve amyloid fibrin.
Beyond their structural resistance, these microclots also trap inflammatory molecules within their matrix. Proteomics analysis by the Pretorius and Kell groups found that Long COVID microclots contain elevated levels of pro-inflammatory proteins, complement activation markers, and von Willebrand factor — creating what amounts to mobile packages of inflammatory material that continuously activate the immune system wherever they circulate.
A 2022 landmark paper by Kell, Laubscher, and Pretorius in the Biochemical Journal formally established microclots as a central driver of Long COVID pathology, noting that these structures persist in the plasma of Long COVID patients even when they are not in an active clotting event. They circulate freely, obstructing capillaries, and sustaining a chronic inflammatory state.
More recent research published in the Journal of Medical Virology (2025) confirmed that microclots in Long COVID patients are structurally associated with neutrophil extracellular traps (NETs), providing another mechanism by which they perpetuate thromboinflammation and immune dysregulation.
The Symptoms Microclotting Produces
The capillary bed is where oxygen, nutrients, and cellular waste products are exchanged between blood and tissues. When microclots obstruct these vessels, the consequences are predictable:
- Fatigue and post-exertional malaise: Reduced oxygen delivery to muscles and mitochondria
- Brain fog and cognitive dysfunction: Impaired cerebral microcirculation
- Breathlessness and exercise intolerance: Reduced pulmonary capillary perfusion
- Small fiber neuropathy and tingling: Nerve tissue hypoxia from microvascular obstruction
- Chest discomfort: Myocardial microvascular involvement
- Temperature dysregulation: Peripheral microcirculatory dysfunction
These are among the most commonly reported and most treatment-resistant symptoms in Long COVID. For patients where microclotting is a significant driver, failing to address it means failing to address a foundational cause of their persistent symptoms. Of course, these symptoms can have other potential driving factors that also must be addressed, such as MCAS, chronic Cell Danger Response, POTS, and more.
PAI-1: The Body’s Clot-Clearing Brake
To understand why some patients are far more vulnerable to persistent microclotting than others, you need to understand a protein called Plasminogen Activator Inhibitor-1, or PAI-1.
PAI-1 is the primary regulator of fibrinolysis, the clot-clearing process. Its job is to inhibit the enzymes (tissue plasminogen activator, or tPA, and urokinase plasminogen activator, or uPA) that convert plasminogen into plasmin, the enzyme that dissolves fibrin. In other words, PAI-1 is the brake on clot dissolution.
This braking function is necessary. The body doesn’t want clots dissolving prematurely when they’re serving a purpose (ie: stopping bleeding). But in a context where amyloid microclots are forming continuously and need to be cleared as quickly as possible, excessive PAI-1 activity is a serious problem. It keeps the brake partially applied when you need full fibrinolytic capacity.
And here’s where genetics becomes directly clinically relevant.
The PAI-1 4G/5G Polymorphism: A Genetic Modifier of Microclotting Risk
In the promoter region of the SERPINE1 gene, the gene that encodes PAI-1, there is a well-characterized genetic variant called the 4G/5G polymorphism. This refers to a single position in the DNA sequence where individuals carry either four consecutive guanosine bases (4G) or five (5G).
This small difference has significant functional consequences for how much PAI-1 your cells produce.
The Three Genotypes
The 4G/5G polymorphism produces three possible genotypes:
4G/4G (homozygous 4G): Both copies of the gene carry the 4G allele. The 5G allele has an additional transcriptional repressor binding site that reduces PAI-1 gene expression — the 4G allele lacks this site. Carrying two 4G alleles means higher baseline PAI-1 production, suppressed fibrinolysis, and significantly elevated thrombosis risk. Under inflammatory conditions, including COVID-19 infection and spike protein exposure, PAI-1 production in 4G/4G individuals ramps up further and is more difficult to suppress.
4G/5G (heterozygous): One copy of each allele. PAI-1 levels are intermediate. Research shows that inflammatory signals like IL-1β still enhance PAI-1 production in 4G/5G endothelial cells, though less dramatically than in 4G/4G individuals.
5G/5G (homozygous 5G): Both copies carry the 5G allele. Lowest baseline PAI-1 production, most active fibrinolysis. However, and this is a crucial nuance, 5G/5G individuals have a different risk profile in COVID-19 contexts. With the fibrinolytic brake released, these patients can develop overactive fibrinolysis and inflammation-driven endothelial dysfunction through a different mechanism.
A 2024 study published in Frontiers in Immunology by Yatsenko, Heissig, and colleagues at Juntendo University confirmed these distinct mechanistic profiles in COVID-19 patients, finding that 4G/4G individuals showed high circulating PAI-1 complexed with plasminogen activators, low plasmin levels, and NF-κB upregulation – a pattern of fibrinolytic shutdown under inflammatory conditions. The 5G/5G group showed the opposite: lower PAI-1, elevated free plasminogen activators, and a profile of inflammation-driven endothelial dysfunction.
Population Prevalence
The 4G allele is common. Population genetics research suggests approximately:
- 25–30% of people carry the 4G/4G genotype
- 50% carry the 4G/5G heterozygous genotype
- 20–25% carry the 5G/5G genotype
This means roughly half to three-quarters of the general population carries at least one 4G allele — and among patients with severe, persistent Long COVID, this proportion may be even higher given the known interaction between the 4G allele and spike protein-driven inflammatory signaling.
Why This Matters Clinically: Two Different Problems, Two Different Approaches
The clinical significance of the 4G/5G polymorphism is not merely academic. It has direct implications for how microclotting should be treated in individual Long COVID and Post-Vaccine Syndrome patients, and why a uniform anticoagulation approach for all patients is inadequate.
The 4G/4G Patient: Fibrinolytic Suppression
For patients with the 4G/4G genotype, the central problem is that spike protein-driven inflammation severely suppresses fibrinolysis. PAI-1 rises under inflammatory conditions, plasmin activity is reduced, and the body’s capacity to clear microclots is significantly impaired.
In these patients, the therapeutic priority is: reduce PAI-1 activity and/or directly enhance fibrinolysis. This is where fibrinolytic enzymes – nattokinase, lumbrokinase – become particularly relevant. Both enzymes work through mechanisms that directly counter the 4G/4G problem.
Nattokinase is a serine protease derived from fermented soybeans (natto) that works through two complementary mechanisms: it directly cleaves fibrin, and it inactivates PAI-1 — the precise molecular target that is overexpressed in 4G/4G patients. Research published in the Journal of Agricultural and Food Chemistry demonstrated that nattokinase directly hydrolyzes PAI-1, increasing fibrinolytic activity. The combination of direct fibrin cleavage and PAI-1 inhibition makes it particularly well-suited to the 4G/4G mechanism.
Lumbrokinase, derived from earthworm species, operates through direct fibrinolytic action and plasminogen activator stimulation. Research by the PolyBio Research Foundation has initiated a clinical trial specifically examining lumbrokinase in Long COVID and ME/CFS, reflecting the growing clinical and mechanistic case for fibrinolytic enzymes in microclot-driven post-viral illness.
Beyond enzymes, sulodexide – a glycosaminoglycan with both anticoagulant and endothelial-protective properties – is used in our practice for patients with evidence of microclotting and endothelial involvement. Unlike systemic anticoagulants, sulodexide has a favorable safety profile for longer-term use and directly supports endothelial repair, which is important given that endothelial dysfunction is both a consequence of microclotting and a driver of further PAI-1 elevation.
The 5G/5G Patient: Inflammation-Driven Endothelial Dysfunction
For patients with the 5G/5G genotype, the problem is different. Fibrinolysis is not suppressed, but inflammation-driven endothelial dysfunction creates a prothrombotic state through other pathways, including elevated uPA and activated complement. These patients may be more prone to systemic inflammation and immune dysregulation than to pure fibrinolytic failure.
The 2024 Juntendo University study specifically identified 5G/5G patients as being at risk for inflammation-induced endothelial dysfunction with fibrinolytic overactivation, a phenotype where aggressive fibrinolytic therapy carries different risk considerations and where anti-inflammatory and endothelial-supportive strategies may be the more appropriate primary focus.
This is clinically significant: the 5G/5G patient who receives high-dose fibrinolytic enzymes without consideration of their genotype is receiving a treatment rationale designed for a different problem. Anticoagulation approach needs to be matched to mechanism, not applied uniformly.
The 4G/5G Patient: Intermediate Risk, Moderate Response
Heterozygous patients show an intermediate profile. The Juntendo study found that IL-1β still enhances PAI-1 production in 4G/5G endothelial cells, though less severely than in 4G/4G. These patients benefit from fibrinolytic support but may need less aggressive dosing and may respond well to lower-intensity anticoagulation combined with strong anti-inflammatory support.
Testing: What We Look For and When
Knowing a patient’s PAI-1 genotype is straightforward. It can be obtained through standard genetic testing including Labcorp’s PAI-1 4G/5G Polymorphism panel (test code 500309).
Beyond genotype, we also look at functional markers of clotting and fibrinolytic activity when clinically indicated:
- PAI-1 functional activity levels (plasma)
- D-dimer (marker of ongoing fibrin degradation — elevated in active microclotting)
- Fibrinogen levels
- Von Willebrand factor antigen (marker of endothelial activation)
- Microclotting levels through fluorescence microscopy
Importantly, standard blood tests and imaging do not detect microclots. The specialized fluorescence microscopy techniques used in Pretorius’s laboratory research are not commercially available at just any lab. We are able to offer this testing to our patients.
This is an area where pattern recognition from clinical experience matters significantly. The presentation of a patient with 4G/4G genotype, elevated D-dimer, elevated fibrinogen, and symptoms strongly suggestive of microvascular obstruction tells a coherent story that guides a different treatment approach than a patient with 5G/5G genotype and primarily inflammatory, autonomic symptoms.
The Broader Picture: Microclotting Doesn’t Operate in Isolation
It’s important to place microclotting in the context of Long COVID’s full complexity. Persistent microclots are a significant mechanism for many patients, but they rarely operate alone. They interact with and compound other pathophysiological drivers:
Microclotting and Cell Danger Response: Tissue hypoxia from microvascular obstruction can itself activate and sustain the Cell Danger Response. Cells detect oxygen insufficiency as a threat and shift into the protective metabolic state we discussed in our CDR post. Addressing microclotting may be a necessary prerequisite to allowing the CDR to resolve in some patients.
Microclotting and POTS/Dysautonomia: Endothelial dysfunction from microclotting directly affects autonomic regulation of vascular tone. Many patients with post-COVID POTS have a vascular endothelial component to their dysautonomia that won’t fully resolve without addressing the underlying endothelial damage.
Microclotting and Neurological Symptoms: The 2025 Journal of Medical Virology study confirmed that microclots in Long COVID are structurally associated with NETs markers, including myeloperoxidase and neutrophil elastase, which can themselves cross the blood-brain barrier and contribute to neuroinflammation. Brain fog in these patients has a partially vascular etiology, not just neurological.
Microclotting and Senescent Cells: Chronic endothelial damage from persistent microclotting can itself drive cellular senescence in vascular endothelial cells, creating a feedback loop where senescent endothelial cells produce SASP-driven pro-inflammatory and pro-thrombotic signals that generate more clotting. This interaction is one reason why microclotting in some patients is difficult to resolve without simultaneously addressing cellular senescence.
The clinical implication is that microclotting treatment is rarely sufficient as a standalone intervention. It typically needs to occur in parallel with reducing the spike protein burden that is driving fibrinogen misfolding, addressing the inflammatory environment that elevates PAI-1, and supporting the endothelial repair that allows normal fibrinolytic function to resume.
A Note on Safety and Clinical Oversight
Fibrinolytic therapy, whether enzymatic or pharmaceutical, requires appropriate clinical supervision. The primary risk is bleeding, and the probability of this risk increases meaningfully when fibrinolytic agents are combined with pharmaceutical anticoagulants (aspirin, clopidogrel, apixaban, warfarin, heparin) without careful monitoring.
This is not an argument against fibrinolytic treatment. We have treated over a thousand patients with triple anticoagulation therapy combined with enzymatic therapies. It is an argument for doing it with proper clinical oversight, appropriate dosing based on individual presentation and genotype, and awareness of the full medication picture. The patients who have the worst outcomes with DIY fibrinolytic protocols are typically those combining multiple agents without understanding their additive effects. Not only that, they are not dealing with the complete picture of spike protein injury.
At Leading Edge Clinic, anticoagulation approach is individualized to the patient’s genetic profile, symptom presentation, functional markers, and complete medication list. For some patients, nattokinase alone at appropriate dosing is the right starting point. For others, sulodexide plays a primary role. For a smaller number of patients with more significant microclotting burden and appropriate clinical indicators, pharmaceutical anticoagulation is warranted. There is no universal protocol.
Conclusion
Fibrin amyloid microclots represent one of the most mechanistically coherent and clinically important, yet most frequently missed, drivers of persistent Long COVID and Post-Vaccine Syndrome symptoms. They explain a cluster of symptoms (fatigue, brain fog, breathlessness, post-exertional malaise) that don’t respond to anti-inflammatories alone because the problem isn’t only inflammation. The problem is physical obstruction of microcirculation and ongoing thromboinflammation.
And the PAI-1 4G/5G polymorphism explains something that purely inflammation-focused frameworks can’t: why patients with similar spike protein exposure and similar inflammatory burdens have dramatically different microclotting trajectories. Your genetics determine how effectively your body can clear these abnormal clots, and they should determine how you treat them.
This is the kind of individualized, mechanism-informed clinical reasoning that drives our approach at Leading Edge Clinic. If you’re experiencing symptoms consistent with microclotting and you haven’t been evaluated for fibrinolytic capacity or PAI-1 genotype, that may be a meaningful gap in your care picture.
You can register here to become a patient.
Leading Edge Clinic specializes in Long COVID, Post-Vaccine Syndrome, and complex post-infectious illness. Our providers treat patients across all 50 states via telehealth. Initial evaluations are 60 minutes.
This article is for educational purposes and does not constitute medical advice. Fibrinolytic therapy carries clinical risks and should be undertaken only with appropriate medical supervision.
Key References
- Kell DB, Laubscher GJ, Pretorius E. A central role for amyloid fibrin microclots in long COVID/PASC: origins and therapeutic implications. Biochem J. 2022;479(4):537–559. https://pmc.ncbi.nlm.nih.gov/articles/PMC8883497/
- Yatsenko T, et al. The influence of 4G/5G polymorphism in the plasminogen-activator-inhibitor-1 promoter on COVID-19 severity and endothelial dysfunction. Front Immunol. 2024;15:1445294. https://pmc.ncbi.nlm.nih.gov/articles/PMC11392769/
- Pretorius E, et al. Persistent clotting protein pathology in Long COVID/PASC is accompanied by increased levels of antiplasmin. Cardiovasc Diabetol. 2021;20:172. https://doi.org/10.1186/s12933-021-01359-7
- Thierry AR, et al. Circulating microclots are structurally associated with neutrophil extracellular traps and their amounts are elevated in Long COVID patients. J Med Virol. 2025;97:e70613. https://pmc.ncbi.nlm.nih.gov/articles/PMC12489976/
- Kruger A, et al. Proteomics of fibrin amyloid microclots in Long COVID/PASC shows many entrapped pro-inflammatory molecules. Cardiovasc Diabetol. 2022;21:190. https://doi.org/10.1186/s12933-022-01623-4
- Kell DB, Khan MA, Pretorius E. Fibrinaloid microclots and atrial fibrillation. Biomedicines. 2024;12:891. https://doi.org/10.3390/biomedicines12040891
- Scientific Reports. Elevated plasma level of PAI-1 is associated with severe COVID-19. Sci Rep. 2025;15:28380. https://doi.org/10.1038/s41598-025-06517-5